Back
PAD: Tibial
034 - Early Experience With a Novel Drug Eluting Stent In the Infrapopliteal Circulation: Initial Results

Peter Faries, MD – Professor-MSH/QHC/ELM, Vascular Surgery, Mount Sinai; Aaron Fischman, MD – Professor-MSH/QHC/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai; Scott Nowakowski, MD – Associate Professor-MSH/QHC/SSVS/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai; Rajesh Patel, MD – Assistant Professor-MSH/MSQ/QHC/SSVS/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai; Dan Shilo, MD – Assistant Professor-MSH/MSQ/QHC/SSVS/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai; Rami Tadros, MD – Professor-MSH/QHC/ELM, Vascular Surgery, Mount Sinai; Ageliki Vouyouka, MD – Professor-MSH/QHC/ELM, Vascular Surgery, Mount Sinai; Vivan Bishay, MD – Assistant Professor-MSH/MSQ/QHC/SSVS/ELM Diagnostic, Molecular & Interventional Radiology, Mount Sinai; Kirema Garcia-Reyes, MD – Assistant Professor-MSH/MSQ/QHC/SSVS/ELM, Mount Sinai; Edward Kim, MD – Associate Professor-MSH/QHC/SSVS/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai; Rahul Patel, MD – Assistant Professor-MSH/MSQ/QHC/SSVS/ELM Diagnostic, Molecular and Interventional Radiology, Mount Sinai
Purpose: Chronic limb-threatening ischemia (CLTI) is a highly morbid, limb and life-threatening, late-stage presentation of peripheral arterial disease (PAD). Below the knee (BTK) intervention of CTLI, while particularly challenging, is an area of rapid development, with constant exploration of new techniques and equipment. The development of drug eluting stents (DES) has significantly impacted the landscape of endovascular revascularization. Initially used for coronary artery stenosis, SYNERGY Everolimus-Eluting Stent (Boston Scientific, Marlborough, MA, USA) employs a novel bioresorbable polymer to enhance the safety and efficacy profile of the stent. The purpose of this study is to review the safety and efficacy and of this novel DES when used to treat infrapopliteal disease in the setting of CLTI.
Material and Methods: All patients who underwent BTK intervention with placement of at least one SYNERGY stent were identified through our electronic medical record system. The data collected includes: Age at procedure, gender, Rutherford category, target vessel occlusion vs stenosis, number of synergy stents placed, stent location, whether there was simultaneous intervention of femoral or popliteal disease, and occurrence of any major adverse event (i.e. above ankle amputation, death, stroke, myocardial infarction (MI)) within 30 days of intervention.
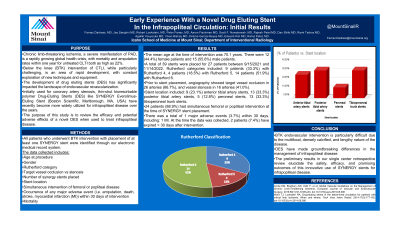
Results: Between Sept 2021 and November 2022, 27 patients were treated with a total of 39 SYNERGY stents. There were 12 female patients and 15 male patients, with mean age of 70.1 at the time of intervention. All stents were placed after failed angioplasty of the target lesion. Rutherford categories included: 9 patients with Rutherford 4, 4 patients with Rutherford 5, 14 patients with Rutherford 6. Prior to stent placement, angiography showed target vessel occlusion in 26 arteries, and vessel stenosis in 13 arteries. Stent location included: 9 anterior tibial artery stents, 13 posterior tibial artery stents, 5 peroneal stents, 13 tibioperoneal trunk stents. 24 patients had simultaneous femoral or popliteal intervention at the time of SYNERGY stent placement. There was a total of 1 major adverse event within 30 days (MI). One planned minor amputation was performed in a Rutherford 6 patient in the first 30 days as well.
Conclusions: These initial results demonstrate the safety and efficacy of this novel drug eluting stent in the treatment of infrapopliteal disease for CLTI. Follow up including limb salvage and target lesion patency will be presented at the conference.
Material and Methods: All patients who underwent BTK intervention with placement of at least one SYNERGY stent were identified through our electronic medical record system. The data collected includes: Age at procedure, gender, Rutherford category, target vessel occlusion vs stenosis, number of synergy stents placed, stent location, whether there was simultaneous intervention of femoral or popliteal disease, and occurrence of any major adverse event (i.e. above ankle amputation, death, stroke, myocardial infarction (MI)) within 30 days of intervention.
Results: Between Sept 2021 and November 2022, 27 patients were treated with a total of 39 SYNERGY stents. There were 12 female patients and 15 male patients, with mean age of 70.1 at the time of intervention. All stents were placed after failed angioplasty of the target lesion. Rutherford categories included: 9 patients with Rutherford 4, 4 patients with Rutherford 5, 14 patients with Rutherford 6. Prior to stent placement, angiography showed target vessel occlusion in 26 arteries, and vessel stenosis in 13 arteries. Stent location included: 9 anterior tibial artery stents, 13 posterior tibial artery stents, 5 peroneal stents, 13 tibioperoneal trunk stents. 24 patients had simultaneous femoral or popliteal intervention at the time of SYNERGY stent placement. There was a total of 1 major adverse event within 30 days (MI). One planned minor amputation was performed in a Rutherford 6 patient in the first 30 days as well.
Conclusions: These initial results demonstrate the safety and efficacy of this novel drug eluting stent in the treatment of infrapopliteal disease for CLTI. Follow up including limb salvage and target lesion patency will be presented at the conference.
