Back
Introduction: The artificial urinary sphincter (AUS) is an effective surgical treatment for stress urinary incontinence, and about 9% of AUS devices require removal due to infection. We sought to determine microbial biofilm and metabolite composition on AUS devices, reconstitute biofilms from microbes isolated from indwelling AUSs in an environment mimicking human tissue, and compare their formation across strains and medical material types. We hypothesized AUSs would harbor unique bacterial and metabolite profiles, and that reconstitution of biofilms in vitro may lead to new strategies to prevent device-associated infection.
Methods: Devices from consented patients scheduled for AUS removal or revision were swabbed for biofilm. Samples and controls were subjected to next-generation sequencing, metabolomics, and culture-based approaches. Bacterial isolates were isolated from devices and grown in a continuous-flow stir-tank bioreactor designed to mimic human tissue with an indwelling medical device present, and biofilm-formation on medical device material types was assessed with plate-count assays and scanning electron microscopy.
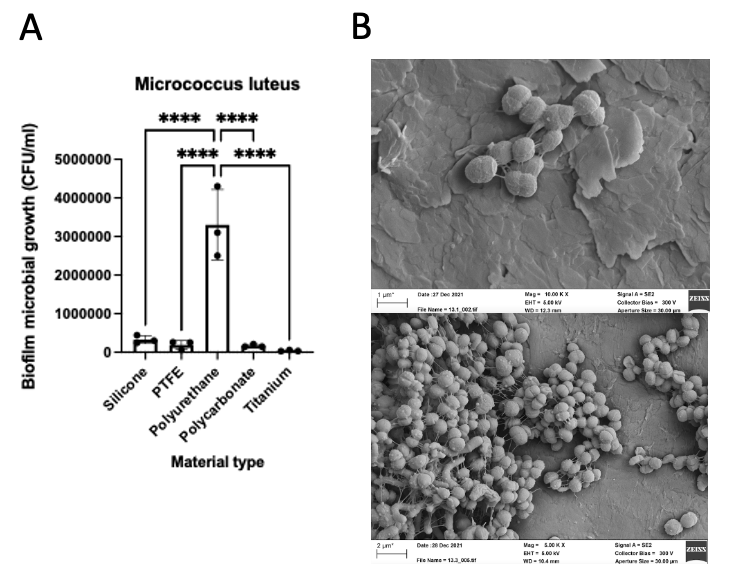
Results: In the 17 analyzed devices, the most commonly detected phyla were Proteobacteria and Firmicutes. Diethanolamine (local anesthetic precursor), threonine-butyl-ester (precursor for peptide synthesis), 5-aminovaleric acid (bacterial metabolite of lysine; GABA agonist) were the most commonly detected metabolites. Microbes isolated from AUS devices consistently reconstituted biofilm in vitro, and biofilm deposition differed by both strain (Staphylococcus lugdunensis the greatest) and material type (polyurethane the greatest and titanium the least, ****p < 0.0001). Micrococcus luteus, shown in the Figure, had intermediate biofilm overall, with silicone shown in the top micrograph and polyurethane on the bottom.
Conclusions: AUS devices consistently harbored microbiota in the form of biofilms. Isolated strains reconstituted biofilm in vitro across a library of medical device materials. Biofilm formation differed by both strain and material type. Our methods and results open new avenues to screen for inhibitors of biofilm formation and novel medical device materials resistant to infection. SOURCE OF
Funding: N/A
Podium Session
Session: PD35: Trauma/Reconstruction/Diversion: Urethral Reconstruction (including Stricture & Diverticulum) I
PD35-01: Artificial urinary sphincter devices harbor microbial biofilms that reconstitute differentially in vitro by strain and medical device material type
Sunday, April 30, 2023
3:30 PM – 3:40 PM CST
Location: S401C

Glenn T. Werneburg, MD, PHD
Cleveland Clinic
Podium Presenter(s)
Introduction: The artificial urinary sphincter (AUS) is an effective surgical treatment for stress urinary incontinence, and about 9% of AUS devices require removal due to infection. We sought to determine microbial biofilm and metabolite composition on AUS devices, reconstitute biofilms from microbes isolated from indwelling AUSs in an environment mimicking human tissue, and compare their formation across strains and medical material types. We hypothesized AUSs would harbor unique bacterial and metabolite profiles, and that reconstitution of biofilms in vitro may lead to new strategies to prevent device-associated infection.
Methods: Devices from consented patients scheduled for AUS removal or revision were swabbed for biofilm. Samples and controls were subjected to next-generation sequencing, metabolomics, and culture-based approaches. Bacterial isolates were isolated from devices and grown in a continuous-flow stir-tank bioreactor designed to mimic human tissue with an indwelling medical device present, and biofilm-formation on medical device material types was assessed with plate-count assays and scanning electron microscopy.
Results: In the 17 analyzed devices, the most commonly detected phyla were Proteobacteria and Firmicutes. Diethanolamine (local anesthetic precursor), threonine-butyl-ester (precursor for peptide synthesis), 5-aminovaleric acid (bacterial metabolite of lysine; GABA agonist) were the most commonly detected metabolites. Microbes isolated from AUS devices consistently reconstituted biofilm in vitro, and biofilm deposition differed by both strain (Staphylococcus lugdunensis the greatest) and material type (polyurethane the greatest and titanium the least, ****p < 0.0001). Micrococcus luteus, shown in the Figure, had intermediate biofilm overall, with silicone shown in the top micrograph and polyurethane on the bottom.
Conclusions: AUS devices consistently harbored microbiota in the form of biofilms. Isolated strains reconstituted biofilm in vitro across a library of medical device materials. Biofilm formation differed by both strain and material type. Our methods and results open new avenues to screen for inhibitors of biofilm formation and novel medical device materials resistant to infection. SOURCE OF
Funding: N/A

