Back
Introduction: Recently, intravesical interferon (IFNa) gene therapy has demonstrated success in treatment of BCG-unresponsive NMIBC in a large, randomized, phase-III trial. While the results have been promising, there is room for improvement. One strategy to improve treatment efficacy and durability may be to identify actionable resistance mechanisms and employ targeted combination therapies.
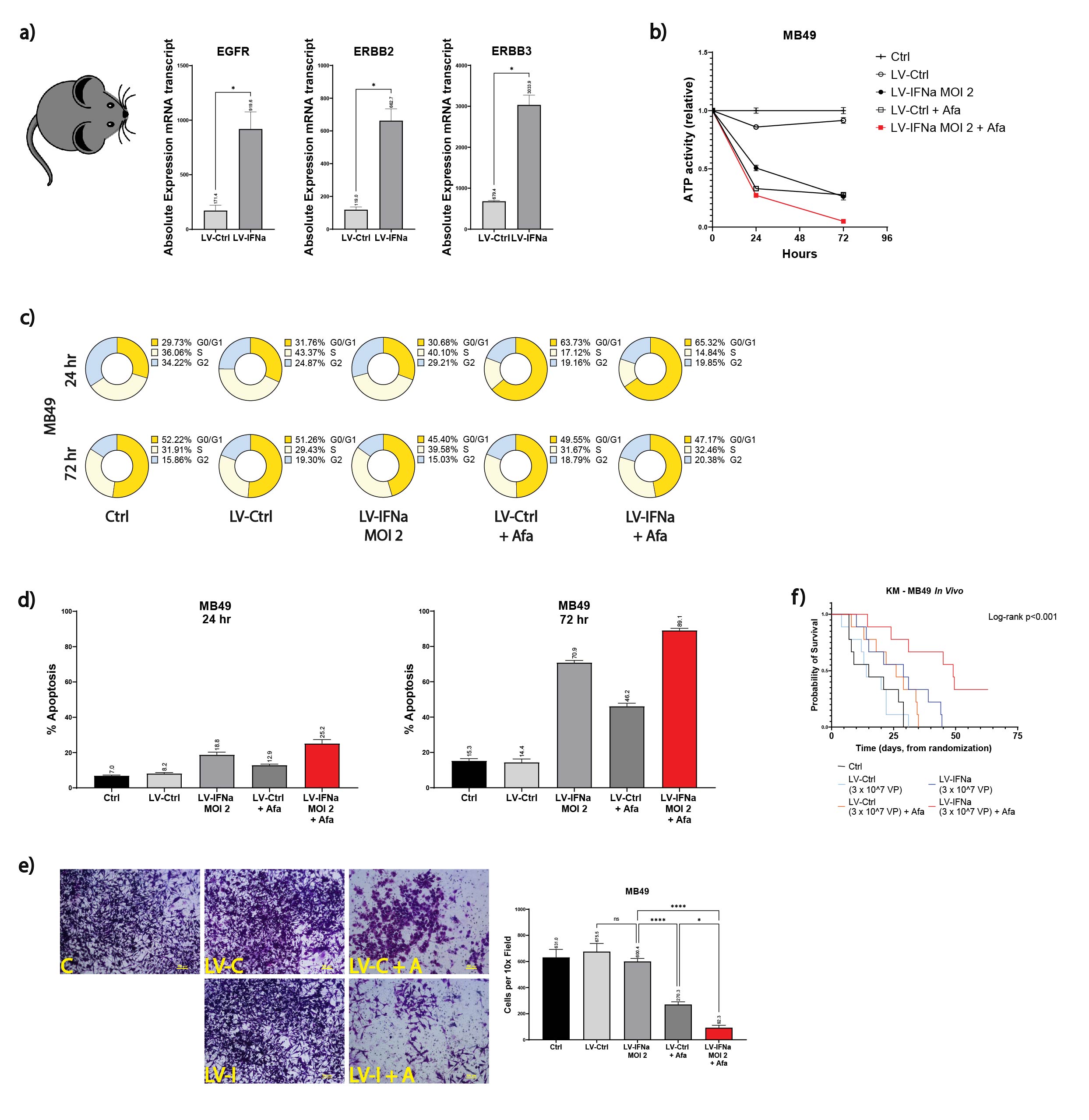
Methods: We performed end-tumor RNA-seq analysis of MB49 mice tumors treated with IFNa gene therapy to identify potential mechanisms of therapy resistance – ErbB pathway was discovered as a potential target (Figure a). We therefore tested combination treatment with ErbB pathway blocker and IFNa in MB49 cells using lentiviral IFNa (LV-IFNa) with/without Afatinib in vitro. Next, in vivo studies were conducted in the MB49 orthotopic mice bladder cancer model: mice were randomized in to 5 treatment groups (n=10 each), saline (Ctrl), LV-Ctrl, Afatinib monotherapy, LV-IFNa monotherapy, and the experimental LV-IFNa + Afatinib combination therapy. Overall survival was assessed.
Results: Combination therapy significantly reduced the viability of MB49 cells in vitro compared to any of the other treatment conditions (mean relative ATPase activity for combination treatment at 72 h 4%, compared to 100%, 26%, and 28% for Ctrl, LV-IFNa, and Afatinib, p<0.001; Figure b). This effect on cell viability appeared to be driven by a combination of additive cytostatic and cytolytic effects (Figure c-d). The combination treatment also decreased cell migration (mean migrated cells/10x field on Boyden chamber assay: 92.3 for combination therapy and 631.0, 600.4, and 270.3 for Ctrl, LV-IFNa, and Afatinib, p<0.001; Figure e). Finally, in vivo studies demonstrated improved OS with combination therapy (median OS 49 d in the combination group vs 15, 29, and 26 d in the Ctrl, LV-IFNa, and Afatinib groups, Log-rank p<0.001; Figure f). No mice in the combination group died of drug toxicity.
Conclusions: Our preliminary investigations suggest that ErbB pathway may be a clinically meaningful resistance mechanism, which when targeted alongside IFNa gene therapy may improve overall treatment efficacy. SOURCE OF
Funding: None
Podium Session
Session: PD25: Bladder Cancer: Basic Research & Pathophysiology II
PD25-06: Combination Treatment With Intravesical Interferon Gene Therapy and Pan-ErbB Receptor Family Blocker Improves Survival In Mice With Bladder Cancer
Sunday, April 30, 2023
7:50 AM – 8:00 AM CST
Location: S404A

Akshay Sood, MD
MD
THE UNIV OF TX M D ANDERSON CANCER CENTER
Podium Presenter(s)
Introduction: Recently, intravesical interferon (IFNa) gene therapy has demonstrated success in treatment of BCG-unresponsive NMIBC in a large, randomized, phase-III trial. While the results have been promising, there is room for improvement. One strategy to improve treatment efficacy and durability may be to identify actionable resistance mechanisms and employ targeted combination therapies.
Methods: We performed end-tumor RNA-seq analysis of MB49 mice tumors treated with IFNa gene therapy to identify potential mechanisms of therapy resistance – ErbB pathway was discovered as a potential target (Figure a). We therefore tested combination treatment with ErbB pathway blocker and IFNa in MB49 cells using lentiviral IFNa (LV-IFNa) with/without Afatinib in vitro. Next, in vivo studies were conducted in the MB49 orthotopic mice bladder cancer model: mice were randomized in to 5 treatment groups (n=10 each), saline (Ctrl), LV-Ctrl, Afatinib monotherapy, LV-IFNa monotherapy, and the experimental LV-IFNa + Afatinib combination therapy. Overall survival was assessed.
Results: Combination therapy significantly reduced the viability of MB49 cells in vitro compared to any of the other treatment conditions (mean relative ATPase activity for combination treatment at 72 h 4%, compared to 100%, 26%, and 28% for Ctrl, LV-IFNa, and Afatinib, p<0.001; Figure b). This effect on cell viability appeared to be driven by a combination of additive cytostatic and cytolytic effects (Figure c-d). The combination treatment also decreased cell migration (mean migrated cells/10x field on Boyden chamber assay: 92.3 for combination therapy and 631.0, 600.4, and 270.3 for Ctrl, LV-IFNa, and Afatinib, p<0.001; Figure e). Finally, in vivo studies demonstrated improved OS with combination therapy (median OS 49 d in the combination group vs 15, 29, and 26 d in the Ctrl, LV-IFNa, and Afatinib groups, Log-rank p<0.001; Figure f). No mice in the combination group died of drug toxicity.
Conclusions: Our preliminary investigations suggest that ErbB pathway may be a clinically meaningful resistance mechanism, which when targeted alongside IFNa gene therapy may improve overall treatment efficacy. SOURCE OF
Funding: None

