Back
Introduction: The most common complication of diabetes is bladder dysfunction which is driven in the female Akita type 1 diabetic mouse by inflammation mediated by the NLRP3 inflammasome. It is unclear how diabetes affects signaling pathways responsible for bladder contractility and if this is mediated by NLRP3. Using a female Akita model that develops a temporal progression from bladder overactivity to underactivity, we investigated effects on the cholinergic signaling pathways. To accomplish this we utilized ex vivo myography to determine how neurotransmitter release from efferent nerve terminals and muscarinic receptor activation is impacted in bladders of non-diabetic and diabetic mice ± the NLRP3 gene.
Methods: Akita mice were crossbred with NLRP3-/- mice to yield non-diabetics and diabetics ± NLRP3 gene. Females were aged to 15 and 30 weeks when respective overactive and underactive bladder phenotypes are evident. For ex vivo myography, bladder strips with intact mucosa were used. Efferent neurotransmitter-mediated contractions were assessed using electric field stimulation (EFS) ± the muscarinic receptor antagonist atropine. Contractility to carbachol, a muscarinic receptor agonist, was also assessed (n=5-10 per group).
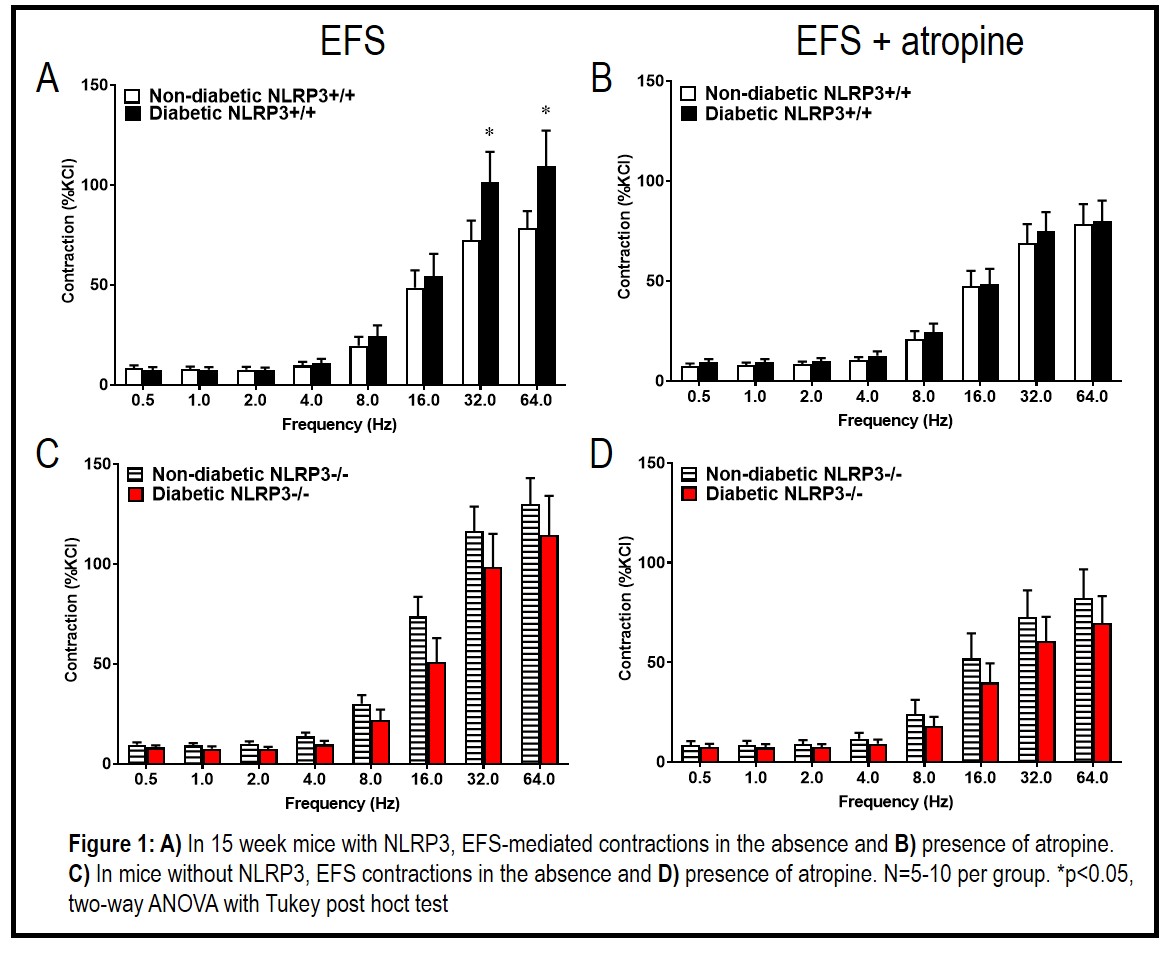
Results: At 15 weeks, diabetic NLRP3+/+ mice have increased EFS contractility (p < 0.05) but NLRP3 gene deletion prevented this pathology in diabetics. At 30 weeks, EFS contractions were no longer dysregulated. EFS was repeated in the presence of atropine to determine the cholinergic contribution. In the presence of atropine, no significant differences were noted in diabetic animals – indicating the increase in contractility at 15 weeks was due to upregulated cholinergic mechanisms. No changes in contractions to carbachol were noted at either time point. This indicates the increased contractility to EFS was not due to increased muscarinic receptor activity but rather an increase in acetylcholine release.
Conclusions: Diabetes increases cholinergic neurotransmitter release and bladder contractility in mice with an overactive bladder as a consequence of inflammation driven by NLRP3. This suggests an NLRP3 inhibitor may be an effective therapy to prevent overactive bladder and eliminate the need for conventional anti-cholinergic treatments. SOURCE OF
Funding: NIH-K12 DK100024; NIH-RO1 DK117890
Podium Session
Session: PD19: Infections/Inflammation/Cystic Disease of the Genitourinary Tract: Kidney & Bladder II
PD19-11: Diabetes causes temporal changes in cholinergic-mediated bladder contractility due to inflammation via the NLRP3 inflammasome in female Akita mice
Saturday, April 29, 2023
2:40 PM – 2:50 PM CST
Location: S401C

Michael Odom, PHD
Postdoctoral Associate
Duke University Medical Center
Podium Presenter(s)
Introduction: The most common complication of diabetes is bladder dysfunction which is driven in the female Akita type 1 diabetic mouse by inflammation mediated by the NLRP3 inflammasome. It is unclear how diabetes affects signaling pathways responsible for bladder contractility and if this is mediated by NLRP3. Using a female Akita model that develops a temporal progression from bladder overactivity to underactivity, we investigated effects on the cholinergic signaling pathways. To accomplish this we utilized ex vivo myography to determine how neurotransmitter release from efferent nerve terminals and muscarinic receptor activation is impacted in bladders of non-diabetic and diabetic mice ± the NLRP3 gene.
Methods: Akita mice were crossbred with NLRP3-/- mice to yield non-diabetics and diabetics ± NLRP3 gene. Females were aged to 15 and 30 weeks when respective overactive and underactive bladder phenotypes are evident. For ex vivo myography, bladder strips with intact mucosa were used. Efferent neurotransmitter-mediated contractions were assessed using electric field stimulation (EFS) ± the muscarinic receptor antagonist atropine. Contractility to carbachol, a muscarinic receptor agonist, was also assessed (n=5-10 per group).
Results: At 15 weeks, diabetic NLRP3+/+ mice have increased EFS contractility (p < 0.05) but NLRP3 gene deletion prevented this pathology in diabetics. At 30 weeks, EFS contractions were no longer dysregulated. EFS was repeated in the presence of atropine to determine the cholinergic contribution. In the presence of atropine, no significant differences were noted in diabetic animals – indicating the increase in contractility at 15 weeks was due to upregulated cholinergic mechanisms. No changes in contractions to carbachol were noted at either time point. This indicates the increased contractility to EFS was not due to increased muscarinic receptor activity but rather an increase in acetylcholine release.
Conclusions: Diabetes increases cholinergic neurotransmitter release and bladder contractility in mice with an overactive bladder as a consequence of inflammation driven by NLRP3. This suggests an NLRP3 inhibitor may be an effective therapy to prevent overactive bladder and eliminate the need for conventional anti-cholinergic treatments. SOURCE OF
Funding: NIH-K12 DK100024; NIH-RO1 DK117890

