Practice Innovations
(PI-004) The Reconstruction of Complex Tissue Defects: The Role of Near Infrared Spectroscopy (NIRS)
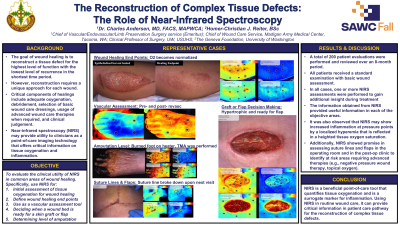
The goal of wound healing is to reconstruct a tissue defect in the shortest period of time, resulting in the highest level of function with the lowest level of recurrence. Reconstruction requires a pathway that is different in every wound. Critical components of the pathway include: assess adequacy of oxygenation to heal the wound, debridement, selection of basic wound care dressings, usage of advanced wound care therapies when required, determination if a wound is healed and prevention of recurrence.
NIRS is an imaging technology that is utilized at point-of-care and can add critical information on tissue oxygenation and inflammation.
Methods:
The following areas were identified as areas that NIRS may add clinically valuable information.
- Initial assessment of adequacy of tissue oxygenation to support wound healing
- Defining when the wound is healed
- Use as vasular assessment tool (inital visit, pre- and post-revascularization)
- Deciding when a wound bed is ready for a skin graft or flap
- Determining level of amputation
Patients were studied to validate the usefulness of NIRS in each area. All patients received a standard history and physical exam and a basic wound assessment. One or more NIRS studies were performed to gain additional information during the course of treatment.
Results: A total of 200 studies were performed and reviewed over a 8 month period. Based on the analysis, the information obtained from the studies provided clinically useful information in each of the areas previously identified.
Additional areas of clinical utility were identified during the project. It was also observed that NIRS may show increased inflammation in pressure points. Additionally, NIRS showed promise in being used to assess suture lines and flaps in the operating room and in post-op clinic to identify areas at risk requiring advanced therapies such as NPWT or topical oxygenation.
Discussion:
NIRS is a beneficial point-of-care tool that measures tissue oxygenation and is a segregate marker for inflammation and can provide critical information in the pathway for the reconstruction of complex tissue defects.
Trademarked Items:
References: 1. U.S. Department of Health and Human Services, Food and Drug Administration Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER), Center for Devices and Radiological Health (CDRH). Guidance for Industry Chronic Cutaneous Ulcer and Burn Wounds — Developing Products for Treatment. June 2006.
2. Snyder D, Sullivan N, Margolis D, Schoelles K. Skin Substitutes for Treating Chronic Wounds. Technology Assessment Program – Technical Brief. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb 2.
3. Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017;376(24):2367-2375.
4. López-Moral M, Lázaro-Martínez JL, García-Morales E, García-Álvarez Y, Álvaro-Afonso FJ, Molines-Barroso RJ. Clinical efficacy of therapeutic footwear with a rigid rocker sole in the prevention of recurrence in patients with diabetes mellitus and diabetic polineuropathy: A randomized clinical trial. PLoS One. 2019;14(7):e0219537.
5. Vedhara K, Beattie A, Metcalfe C, et al. Development and preliminary evaluation of a psychosocial intervention for modifying psychosocial risk factors associated with foot re-ulceration in diabetes. Behav Res Ther. 2012;50(5):323-332.
6. Shahin TB, Vaishnav KV, Watchman M, et al. Tissue augmentation with allograft adipose matrix for the diabetic foot in rem Surg Glob Open. 2017;5(10):e1555.
7. Nickerson DS. Reconsidering nerve decompression: an overlooked opportunity to limit diabetic foot ulcer recurrence and amputation. J Diabetes Sci Technol. 2013;7(5):1195-1201.
8. Najafi B, Reeves ND, Armstrong DG. Leveraging smart technologies to improve the management of diabetic foot ulcers and extend ulcer-free days in remission. Diabetes Metab Res Rev. 2020;36(Suppl 1):e3239.
9. Adiewere P, Gillis RB, Imran Jiwani S, Meal A, Shaw I, Adams GG. A systematic review and meta-analysis of patient education in preventing and reducing the incidence or recurrence of adult diabetes foot ulcers (DFU). Heliyon. 2018;4(5):e00614.
10. LEVENSON SM, GEEVER EF, CROWLEY LV, OATES JF 3rd, BERARD CW, ROSEN H. THE HEALING OF RAT SKIN WOUNDS. Ann Surg. 1965 Feb;161(2):293-308. doi: 10.1097/00000658-196502000-00019. PMID: 14260029; PMCID: PMC1408940.

.png)