Back
Regular Abstract
Therapeutics
Session: Poster Session
18: Post Hoc Analysis of the CLEAR Study in Advanced Renal Cell Carcinoma (aRCC): Subsequent Therapy and Survival Outcomes with Lenvatinib (L) + Everolimus (E) vs Sunitinib (S)
Location: Poster Hall, Board D1
Howard Gurney, MB, BS; Hilary Glen, MbChB, MSc, PhD, FRCP; Robert Motzer, MD; Toni Choueiri, MD; Sun Young Rha, MD; Anna Alyasova, MD; Jamie Merchan, MD; Avivit Peer, MD; Toshio Takagi, MD; Camillo Porta, MD; Viktor Grünwald, MD; Ugo De Giorgi, MD; Ulka Vaishampayan, MD; Manuela Schmidinger, MD; Karla Rodriguez-Lopez, MD; Masatoshi Eto, MD; Lea Dutta, PharmD; Dongyuan Xing, PhD; Thomas Powles
- TH
Thomas E. Hutson, DO, PharmD, FACP
Texas Oncology
Dallas, Texas, United States of America
Poster Presenter(s)
Background: In the 3-arm phase 3 CLEAR study, L+E significantly improved PFS and ORR vs S in 1L aRCC. OS difference for L+E vs S was not statistically significant. We examine the impact of subsequent medication on OS.
Methods: Post hoc analyses examined OS by subgroup in L+E and S arms (data cutoff: August 28, 2020). The 2-stage estimation approach was utilized to adjust for the confounding effect of subsequent anticancer medication for comparison of OS in L+E vs S arms.
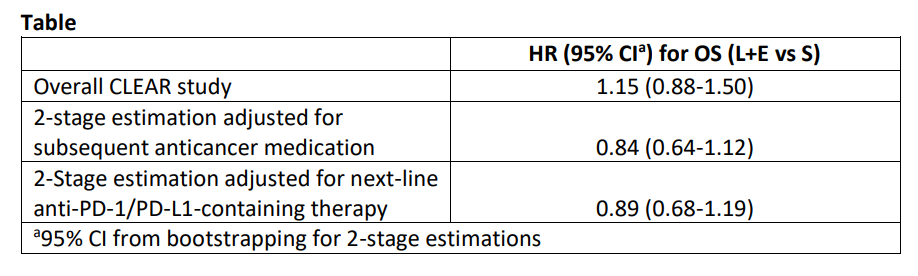
Results: Among randomized patients, 357 were assigned to L+E and S, each. Median duration of follow-up in L+E and S arms was 27 and 26 months, respectively. Given the shorter median duration of treatment with S vs L+E (7.8 vs 11.0 months), more patients in the S arm received subsequent anticancer medication during follow-up (L+E, n=167; S, n=206). Among patients who received subsequent anticancer medication, those in the L+E arm had a longer median time from randomization to initiation of subsequent medication vs those in the S arm (8.0 vs 6.6 months). Analyses of OS by 2-stage estimation adjusted for subsequent anticancer medication are in the table. In the US population subgroup (L+E, n=62; S, n=61), similar numbers of patients received subsequent anticancer medication in the L+E vs S arms (62.9% vs 65.6%), and OS was comparable (HR 0.95, 95%CI 0.51-1.76). Overall, safety was consistent with known safety profiles. Most treatment-emergent deaths were due to progressive disease, not study treatment, with no clustering of events.
Conclusions: In CLEAR, L+E significantly improved PFS vs S (primary endpoint). Exploratory analyses suggest subsequent anticancer medication affected the OS outcome results for L+E vs S.
Methods: Post hoc analyses examined OS by subgroup in L+E and S arms (data cutoff: August 28, 2020). The 2-stage estimation approach was utilized to adjust for the confounding effect of subsequent anticancer medication for comparison of OS in L+E vs S arms.
Results: Among randomized patients, 357 were assigned to L+E and S, each. Median duration of follow-up in L+E and S arms was 27 and 26 months, respectively. Given the shorter median duration of treatment with S vs L+E (7.8 vs 11.0 months), more patients in the S arm received subsequent anticancer medication during follow-up (L+E, n=167; S, n=206). Among patients who received subsequent anticancer medication, those in the L+E arm had a longer median time from randomization to initiation of subsequent medication vs those in the S arm (8.0 vs 6.6 months). Analyses of OS by 2-stage estimation adjusted for subsequent anticancer medication are in the table. In the US population subgroup (L+E, n=62; S, n=61), similar numbers of patients received subsequent anticancer medication in the L+E vs S arms (62.9% vs 65.6%), and OS was comparable (HR 0.95, 95%CI 0.51-1.76). Overall, safety was consistent with known safety profiles. Most treatment-emergent deaths were due to progressive disease, not study treatment, with no clustering of events.
Conclusions: In CLEAR, L+E significantly improved PFS vs S (primary endpoint). Exploratory analyses suggest subsequent anticancer medication affected the OS outcome results for L+E vs S.

