Back
Poster, Podium & Video Sessions
MP01: Benign Prostatic Hyperplasia: Surgical Therapy & New Technology I
MP01-06: Office Based Transperineal Laser Ablation for Benign Prostatic Hyperplasia: 3 & 6 months outcomes.
Friday, May 13, 2022
7:00 AM – 8:15 AM
Location: Room 228
Luna Eusebio*, Luis Avila, Alberto Lopez-Prieto, Edward Gheiler, Ariel Kaufman, David Cohen, Fernando Bianco, Miami, FL
- EL
Poster Presenter(s)
Introduction: BPH affects tens of millions of men across the world. Most procedures require either general or regional anesthesia or a transurethral approach. Herein, we present the 3 & 6 months results of NCT04760483 is a phase I prospective, single center, interventional pilot study evaluating transperineal laser ablation (TPLA) of BPH tissues, carried in Office setting under local anesthesia. A detailed step by step video depiction of this procedure is available at the AUA video library. The objectives call for safety, feasibility, and impact in pertinent outcomes measures, such as Uroflowmetry, IPSS, Hematuria, Erectile function, and ejaculation
Methods: The study contemplated accrual of 20 men between 50 and 80 years with prostate volumes between 30 and 120 cc, IPSS scores >9, peak flows between 5 and 15 cc/s and void residuals under <250 ml. Any patient neurological conditions, history of any surgical intervention or urinary retention were excluded. IPSS assessments, Flow studies and prostate volume measures were conducted at 3 months. Herein we present the results. Bayesian analysis for continuous measurements were performed and non-parametric differences were evaluated using chi2 tests.
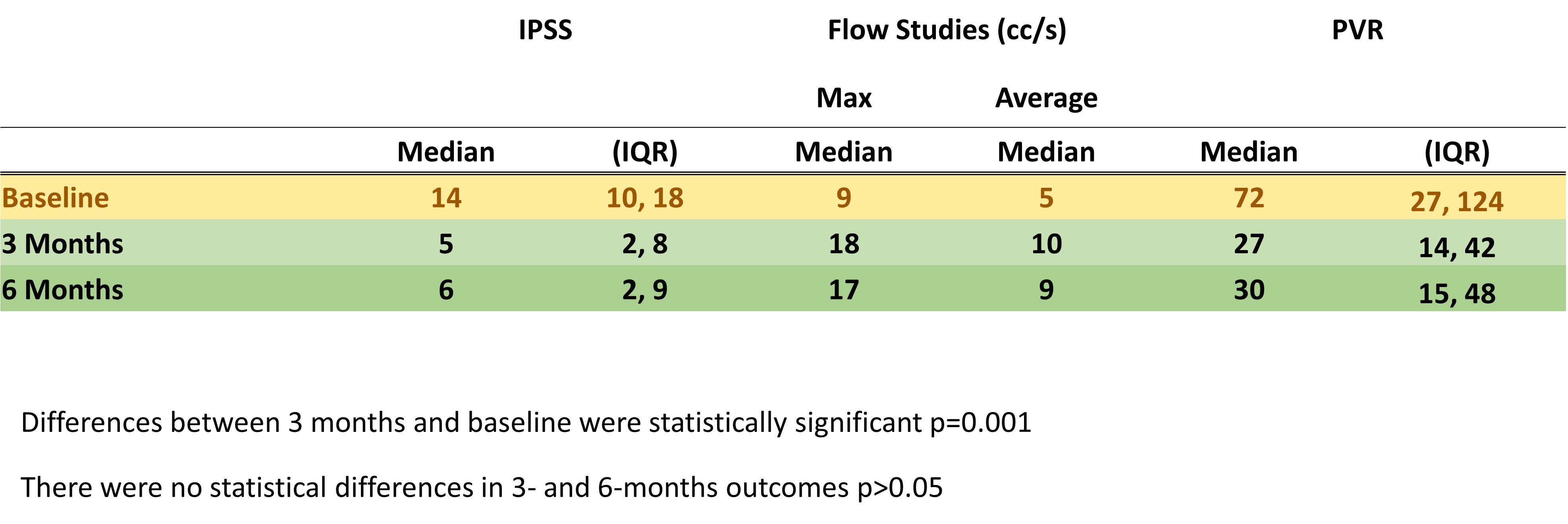
Results: Patients enrolled between December 2020 and February of 2021. The median (IQR) for age and BMI was 68 (58,73) and 29 (27,31), respectively. These parameters for room time, ablation time, watts and total joules were 29 (23,32), 9 minutes (7,12), 6 (5,7) watts and 3,400 (2,600, 3600) joules, respectively. 8(40%) were discharged with a Foley due to elevated residuals. 16 patients had erections and ejaculations before and 3 months after TPLA. 17/20 (85%) had significant improvement in their urinary profile after TPLA (See TABLE for details). One of the initial responders suffered from COVID-19 infection and developed a CVA that hindered his urinary function.
Conclusions: TPLA in the office setting is feasible and safe. Three month outcomes showed subjective and objective sustained improvement in over 80% of patients for at least 6 months. Furthermore, erections or ejaculations were not affected. This novel and promising approach demands further evaluation in phase II-III trials.
Source of Funding: Urological Research Network Foundation
Methods: The study contemplated accrual of 20 men between 50 and 80 years with prostate volumes between 30 and 120 cc, IPSS scores >9, peak flows between 5 and 15 cc/s and void residuals under <250 ml. Any patient neurological conditions, history of any surgical intervention or urinary retention were excluded. IPSS assessments, Flow studies and prostate volume measures were conducted at 3 months. Herein we present the results. Bayesian analysis for continuous measurements were performed and non-parametric differences were evaluated using chi2 tests.
Results: Patients enrolled between December 2020 and February of 2021. The median (IQR) for age and BMI was 68 (58,73) and 29 (27,31), respectively. These parameters for room time, ablation time, watts and total joules were 29 (23,32), 9 minutes (7,12), 6 (5,7) watts and 3,400 (2,600, 3600) joules, respectively. 8(40%) were discharged with a Foley due to elevated residuals. 16 patients had erections and ejaculations before and 3 months after TPLA. 17/20 (85%) had significant improvement in their urinary profile after TPLA (See TABLE for details). One of the initial responders suffered from COVID-19 infection and developed a CVA that hindered his urinary function.
Conclusions: TPLA in the office setting is feasible and safe. Three month outcomes showed subjective and objective sustained improvement in over 80% of patients for at least 6 months. Furthermore, erections or ejaculations were not affected. This novel and promising approach demands further evaluation in phase II-III trials.
Source of Funding: Urological Research Network Foundation


.jpg)
.jpg)