Back
Poster Session A
Vasculitis
Session: (0432–0457) Vasculitis – ANCA-Associated Poster I: Epidemiology, Outcomes, and Classification
0443: Prognosis of Non-PR3 ANCA-Associated Vasculitis with Glomerulonephritis
Saturday, November 12, 2022
1:00 PM – 3:00 PM Eastern Time
Location: Virtual Poster Hall
- AC
Aurélien Chepy, MD
INFINTE U186 inserm, univeristy of Lille
Lille, France
Abstract Poster Presenter(s)
Aurélien Chepy1, Hélène Béhal2, Alexandre Karras3, Xavier Puéchal4, Benjamin Terrier4, David Jayne5, Thomas Quéméneur6 and Mary-Jane Guerry7, 1CHU Lille, Département de Médecine Interne et Immunologie Clinique, Centre de Référence des Maladies Auto-immunes Systémiques Rares du Nord et Nord-Ouest de France (CeRAINO), Lille, France, 2University of Lille, CHU Lille, ULR 2694 - METRICS: Évaluation des Technologies de Santé et des Pratiques Médicales, Lille, France, 3HEGP, Paris, France, 4National Referral Center for Rare Systemic Autoimmune Diseases, Cochin Hospital, Paris, France, 5University of Cambridge, Cambridge, United Kingdom, 6Valenciennes Hospital, Valenciennes, France, 7Department of Nephrology, Centre Hospitalier de Valenciennes, Valenciennes., Valenciennes, France
Background/Purpose: Immunosuppressive treatments have improved the prognosis of anti-neutrophil cytoplasm antibody (ANCA)-associated vasculitis (AAV), transforming these formerly fatal diseases into chronic conditions, with periods of remission and relapse.
Relapse prevention is based on remission-maintenance immunosuppressive treatment with a usual duration of 18 to 24 months. However, prolonged maintenance treatment may not be necessary for all patients. On the one hand, MPO-ANCA (vs PR3-ANCA) and decreased estimated glomerular filtration rate (eGFR) have been associated with a low risk of relapse. On the other hand, treatment of AAV is not devoid of risk. Thus, it has been well demonstrated that patients with renal AAV without PR3 ANCA have the lowest risk of relapse and the highest mortality rate.
This study aimed to determine: first, the risk of late relapse in AAV patients with renal involvement and without PR3-ANCA after end of remission-maintenance treatment, and second, risk factors for relapse in this population.
Methods: Patients with renal involvement without PR3-ANCA recruited to prospective, randomised trials of the European Vasculitis Study group (EUVAS): CYCAZAREM, CYCLOPS, MEPEX, IMPROVE and the French Vasculitis Study Group (FVSG): WEGENT were included in the analysis if their disease was in remission at the end of relapse-prevention therapy. This consisted in azathioprine, methotrexate or mycophenolate mofetil and was stopped after 18-24 months (around 24-27 months after diagnosis).
Results: One hundred and eightpatients were included, 64 of whom (59.3%) were male, 79 were MPO-ANCA positive (73.1%) and 29 ANCA negative (26.9%). Average age at diagnosis was 59.6 +/- 12.4 years. Relapse rate was 25.5% 60 months after cessation of maintenance treatment (figure 1) of which 46.2% were renal relapses (figure 2). Lower GFR at diagnosis was correlated with a lower risk of relapse. For every 15-point decrease in GFR at diagnosis, the risk of relapse decreased by a factor of 0.72 (HR 95% CI: 0.59 to 0.88). Figure 3 represents the cumulative incidence of relapse by eGFR level. Relapse risk did not appear to be impacted by the presence at diagnosis of systemic/constitutional (HR: 0.70; CI 95%: 0.30-1.65; p: 0.47), lung (HR: 0.87; CI 95%: 0.40-1.86; p: 0.71), skin involvement (HR: 0.84;CI 95%: 0.29-2.39; p: 0.74), ear, nose and throat (HR: 1.53; CI 95%: 0.74-3.27; p: 0.28) or eye disease (HR: 1.58; CI 95%: 0.62-4.10; p: 0.34). Presence of MPO-ANCA at diagnosis did not influence relapse-risk.
Conclusion: Even in this population of renal AAV without PR3-ANCA, there remains a risk of late relapse, after the end of a two-year conventional maintenance regimen. Using additional criteria (such as eGFR at diagnosis < or > 45 ml/min/1.73m2) could help select a group of patients who are at lower risk of relapse. Factors that potentially increase (ENT or eye disease) risk of relapse need to be explored further. If these data are confirmed, sub-groups could be identified who could receive only a short maintenance treatment, or perhaps none. These findings also need to be to be analysed following rituximab maintenance.
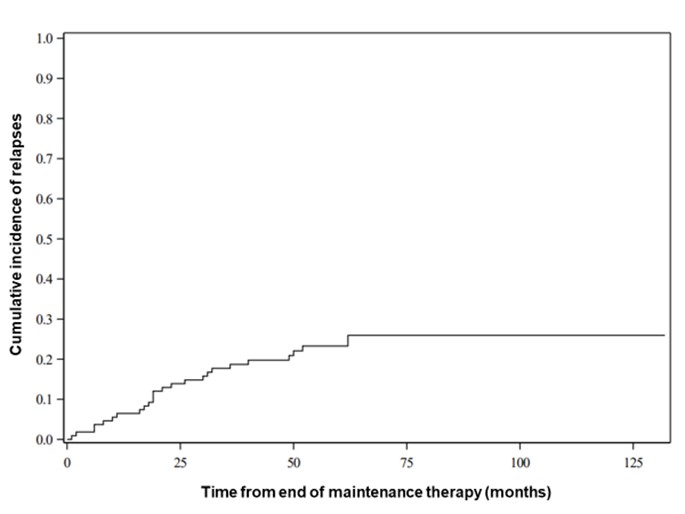 Cumulative incidence of relapse over time
Cumulative incidence of relapse over time
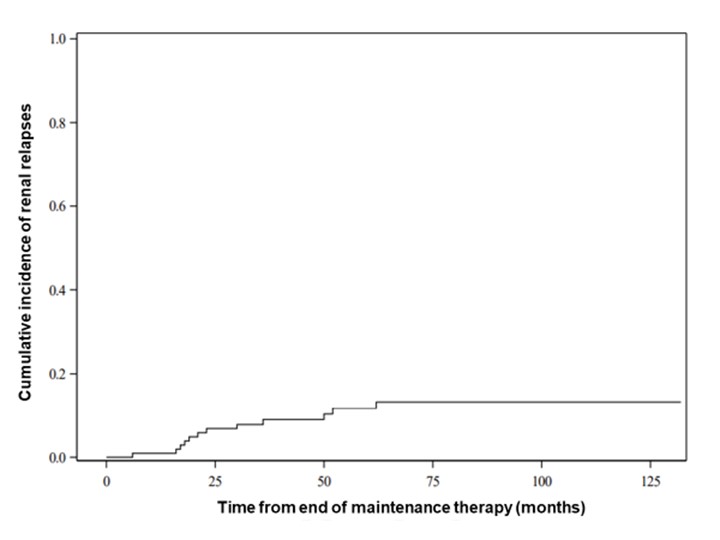 Cumulative incidence of renal relapse over time
Cumulative incidence of renal relapse over time
<img src=https://www.abstractscorecard.com/uploads/Tasks/upload/17574/QHOPTGBB-1289944-3-ANY(1).jpg width=440 height=329.839883551674 border=0 style=border-style: none;>
Disclosures: A. Chepy, None; H. Béhal, None; A. Karras, Roche, Gilead, Amgen; X. Puéchal, Roche; B. Terrier, AstraZeneca, GlaxoSmithKline, Bristol-Myers Squibb(BMS), Eli Lilly, LFB, Boehinger Ingelheim, Vifor Pharma, Pfizer, Roche; D. Jayne, Aurinia, AstraZeneca, GlaxoSmithKline (GSK), Roche/Genentech, Vifor, Bristol-Myers Squibb(BMS), Chemocentryx, Novartis, Takeda, Boehringer-Ingelheim, Otsuka, UCB, Amgen, Kessai; T. Quéméneur, Roche; M. Guerry, None.
Background/Purpose: Immunosuppressive treatments have improved the prognosis of anti-neutrophil cytoplasm antibody (ANCA)-associated vasculitis (AAV), transforming these formerly fatal diseases into chronic conditions, with periods of remission and relapse.
Relapse prevention is based on remission-maintenance immunosuppressive treatment with a usual duration of 18 to 24 months. However, prolonged maintenance treatment may not be necessary for all patients. On the one hand, MPO-ANCA (vs PR3-ANCA) and decreased estimated glomerular filtration rate (eGFR) have been associated with a low risk of relapse. On the other hand, treatment of AAV is not devoid of risk. Thus, it has been well demonstrated that patients with renal AAV without PR3 ANCA have the lowest risk of relapse and the highest mortality rate.
This study aimed to determine: first, the risk of late relapse in AAV patients with renal involvement and without PR3-ANCA after end of remission-maintenance treatment, and second, risk factors for relapse in this population.
Methods: Patients with renal involvement without PR3-ANCA recruited to prospective, randomised trials of the European Vasculitis Study group (EUVAS): CYCAZAREM, CYCLOPS, MEPEX, IMPROVE and the French Vasculitis Study Group (FVSG): WEGENT were included in the analysis if their disease was in remission at the end of relapse-prevention therapy. This consisted in azathioprine, methotrexate or mycophenolate mofetil and was stopped after 18-24 months (around 24-27 months after diagnosis).
Results: One hundred and eightpatients were included, 64 of whom (59.3%) were male, 79 were MPO-ANCA positive (73.1%) and 29 ANCA negative (26.9%). Average age at diagnosis was 59.6 +/- 12.4 years. Relapse rate was 25.5% 60 months after cessation of maintenance treatment (figure 1) of which 46.2% were renal relapses (figure 2). Lower GFR at diagnosis was correlated with a lower risk of relapse. For every 15-point decrease in GFR at diagnosis, the risk of relapse decreased by a factor of 0.72 (HR 95% CI: 0.59 to 0.88). Figure 3 represents the cumulative incidence of relapse by eGFR level. Relapse risk did not appear to be impacted by the presence at diagnosis of systemic/constitutional (HR: 0.70; CI 95%: 0.30-1.65; p: 0.47), lung (HR: 0.87; CI 95%: 0.40-1.86; p: 0.71), skin involvement (HR: 0.84;CI 95%: 0.29-2.39; p: 0.74), ear, nose and throat (HR: 1.53; CI 95%: 0.74-3.27; p: 0.28) or eye disease (HR: 1.58; CI 95%: 0.62-4.10; p: 0.34). Presence of MPO-ANCA at diagnosis did not influence relapse-risk.
Conclusion: Even in this population of renal AAV without PR3-ANCA, there remains a risk of late relapse, after the end of a two-year conventional maintenance regimen. Using additional criteria (such as eGFR at diagnosis < or > 45 ml/min/1.73m2) could help select a group of patients who are at lower risk of relapse. Factors that potentially increase (ENT or eye disease) risk of relapse need to be explored further. If these data are confirmed, sub-groups could be identified who could receive only a short maintenance treatment, or perhaps none. These findings also need to be to be analysed following rituximab maintenance.
 Cumulative incidence of relapse over time
Cumulative incidence of relapse over time Cumulative incidence of renal relapse over time
Cumulative incidence of renal relapse over time<img src=https://www.abstractscorecard.com/uploads/Tasks/upload/17574/QHOPTGBB-1289944-3-ANY(1).jpg width=440 height=329.839883551674 border=0 style=border-style: none;>
Cumulative incidence of relapse by eGFR level (1: < 15 / 2 :15-30 / 3 :30-45 / 4:>=45 ml/min/1.73m2)
Disclosures: A. Chepy, None; H. Béhal, None; A. Karras, Roche, Gilead, Amgen; X. Puéchal, Roche; B. Terrier, AstraZeneca, GlaxoSmithKline, Bristol-Myers Squibb(BMS), Eli Lilly, LFB, Boehinger Ingelheim, Vifor Pharma, Pfizer, Roche; D. Jayne, Aurinia, AstraZeneca, GlaxoSmithKline (GSK), Roche/Genentech, Vifor, Bristol-Myers Squibb(BMS), Chemocentryx, Novartis, Takeda, Boehringer-Ingelheim, Otsuka, UCB, Amgen, Kessai; T. Quéméneur, Roche; M. Guerry, None.

