Back
Poster Session D
Session: (1924–1949) Pediatric Rheumatology – Clinical Poster III: Other
1929: Outcomes and Safety of Anakinra for the Treatment of Multisystem Inflammatory Syndrome in Children
Monday, November 14, 2022
1:00 PM – 3:00 PM Eastern Time
Location: Virtual Poster Hall
- CR
Christopher Redmond, MD, MSc
National Institutes of Health
Rockville, MD, United States
Abstract Poster Presenter(s)
Dizon Brian1, Christopher Redmond2, Emily Gotschlich3, Sangeeta Sule4, Tova Ronis4, Kathleen Vazzana5, Matthew Sherman6, Rachael Connor7, Abigail Bosk4, Niti Dham4, Ashraf Harahsheh4, Elizabeth Wells4, Roberta DeBiasi4 and Hemalatha Srinivasalu4, 1National Institutes of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH), Bethesda, MD, 2National Institutes of Health, Rockville, MD, 3FDA, Washington, DC, 4Children's National Hospital, Washington, DC, 5Arnold Palmer Hospital for Children, Orlando, FL, 6Muscle Disease Unit, Laboratory of Muscle Stem Cells and Gene Regulation, National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), National Institutes of Health (NIH); Children’s National Hospital, Washington, DC, 7Children's National Medical Center, Washington, DC
Background/Purpose: Multisystem inflammatory syndrome in children (MIS-C) is a clinical entity distinct from primary COVID-19 infection that resembles Kawasaki disease (KD) and toxic shock syndrome. Consensus treatment guidelines for MIS-C have been established based on expert review. Anakinra has been used in refractory cases of MIS-C, but has not been studied in-depth.
Methods: A single center retrospective cohort study was conducted of all MIS-C patients hospitalized at CNH from May 15 to November 15, 2020. Diagnostic and therapeutic data were extracted from the electronic health record. Forty-six patients met the CDC case definition for MIS-C and were further stratified by the presence/absence of KD-like features and CV shock. MIS-C patients with neither KD-like features nor CV shock were termed non-specific, while those with both were termed Kawasaki disease shock syndrome (KDSS).
Results: Of the 46 MIS-C patients included in the study, all received IVIG, 14 (30%) received IVIG monotherapy and 32 (70%) received IVIG+anakinra. No mortality was observed, and no statistically significant differences in adverse reactions were found. The median initial dose of anakinra was 7.9 mg/kg/day to a maximum dose of 9.5 mg/kg/day, with a duration of 9.8 days. Most CV shock patients received anakinra (+anakinra 63% versus -anakinra 21%, p=0.02), which were prescribed at higher doses (+CV shock 9.8 mg/kg/day versus -CV shock 8.0 mg/kg/day, p=0.003) and for a longer duration (+CV shock 10.6 days versus -CV shock 7.1 days, p=0.002). In patients who received IVIG+anakinra, >50% defervesced by day 2 of treatment (figure 1A-C). Among patients receiving IVIG+anakinra, KD-like symptoms was associated with a prolonged time to defervescence (figure 1B). MIS-C patients treated with IVIG+anakinra showed improvement in LVEF (59.0% versus follow-up 62.5%, p=0.004) (figure 1D) and Ap4 cardiac strain (median initial -13.2 versus follow-up -17.8, p=0.04) (figure 1E). While improvements in LVEF with IVIG+anakinra remained significant in CV shock and KDSS subgroup analysis (figure 1D and figure 2C), improvement in cardiac strain was significant in those with KD-like features (median initial -3.2 versus follow-up -19.1, p=0.006) (figure 1D), but not CV shock (p=0.37) (figure 1D and 2D).
Conclusion: Treatment of MIS-C with anakinra was associated with resolution of symptoms, including fever and cardiovascular dysfunction, with no significant adverse events. Our findings suggest an alternative strategy associated with resolution of MIS-C-associated hyperinflammation, and may be useful in MIS-C patients for whom steroids may be unfavorable or contraindicated.
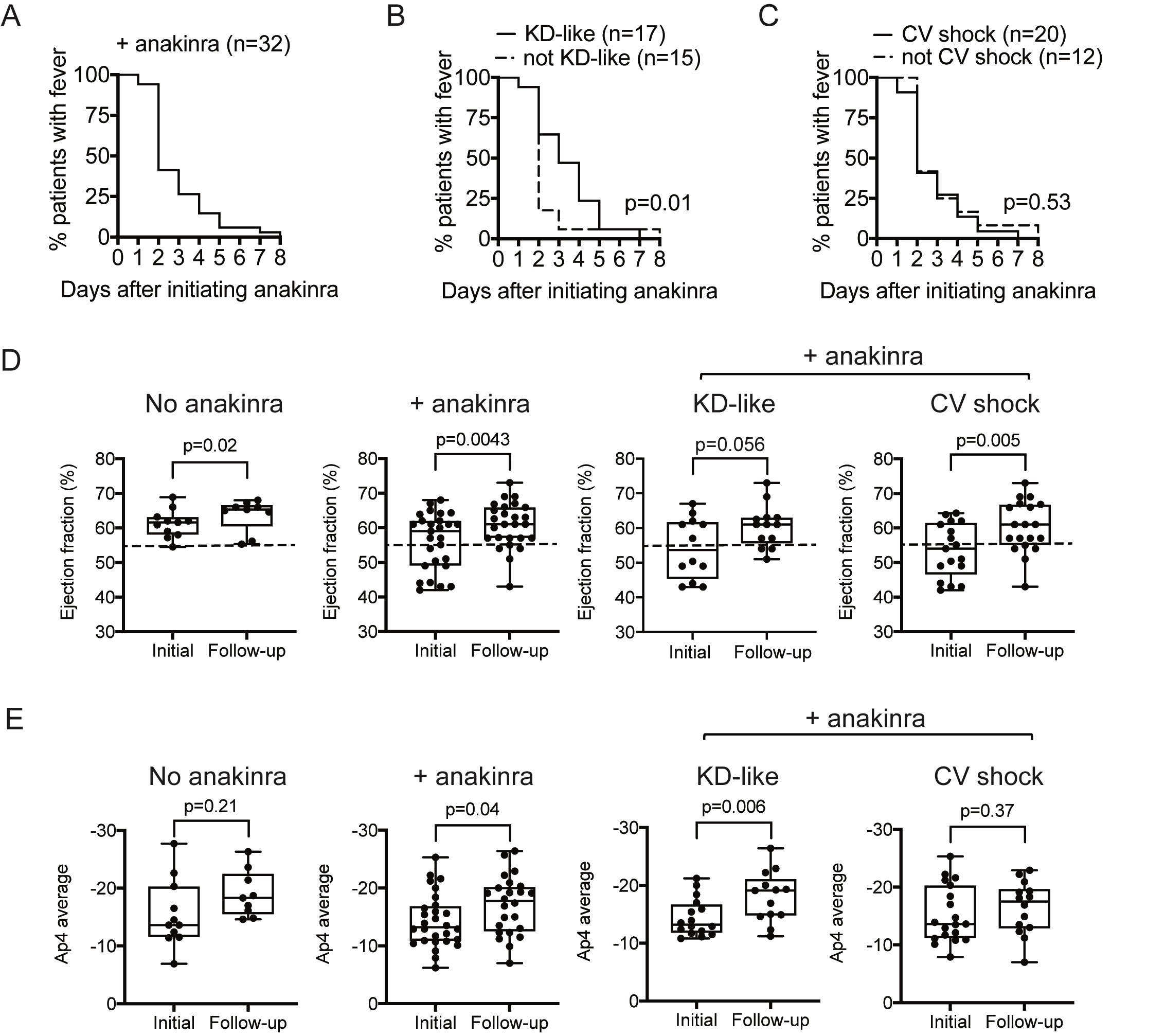 Primary outcome measurements of MIS-C patients with non-specific, KD-like, and CV shock clinical phenotypes treated with anakinra. The effect of anakinra on defervescence in all patients receiving anakinra (A), as well as those with KD-like features (B) and CV shock phenotypes (C), is shown. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as the LVEF of MIS-C patients with KD-like features or CV shock who received anakinra, is shown in D. The lower limit of normal LVEF (55%) is indicated by the dotted lines. Ap4 cardiac strain on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as MIS-C patients with KD-like features or CV shock who had received anakinra, is shown in E. Data were analyzed for statistical significance by log rank test (B and C) or non-parametric two-tailed Wilcoxon matched pairs signed rank test (D and E), and a p-value < 0.05 was considered statistically significant.
Primary outcome measurements of MIS-C patients with non-specific, KD-like, and CV shock clinical phenotypes treated with anakinra. The effect of anakinra on defervescence in all patients receiving anakinra (A), as well as those with KD-like features (B) and CV shock phenotypes (C), is shown. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as the LVEF of MIS-C patients with KD-like features or CV shock who received anakinra, is shown in D. The lower limit of normal LVEF (55%) is indicated by the dotted lines. Ap4 cardiac strain on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as MIS-C patients with KD-like features or CV shock who had received anakinra, is shown in E. Data were analyzed for statistical significance by log rank test (B and C) or non-parametric two-tailed Wilcoxon matched pairs signed rank test (D and E), and a p-value < 0.05 was considered statistically significant.
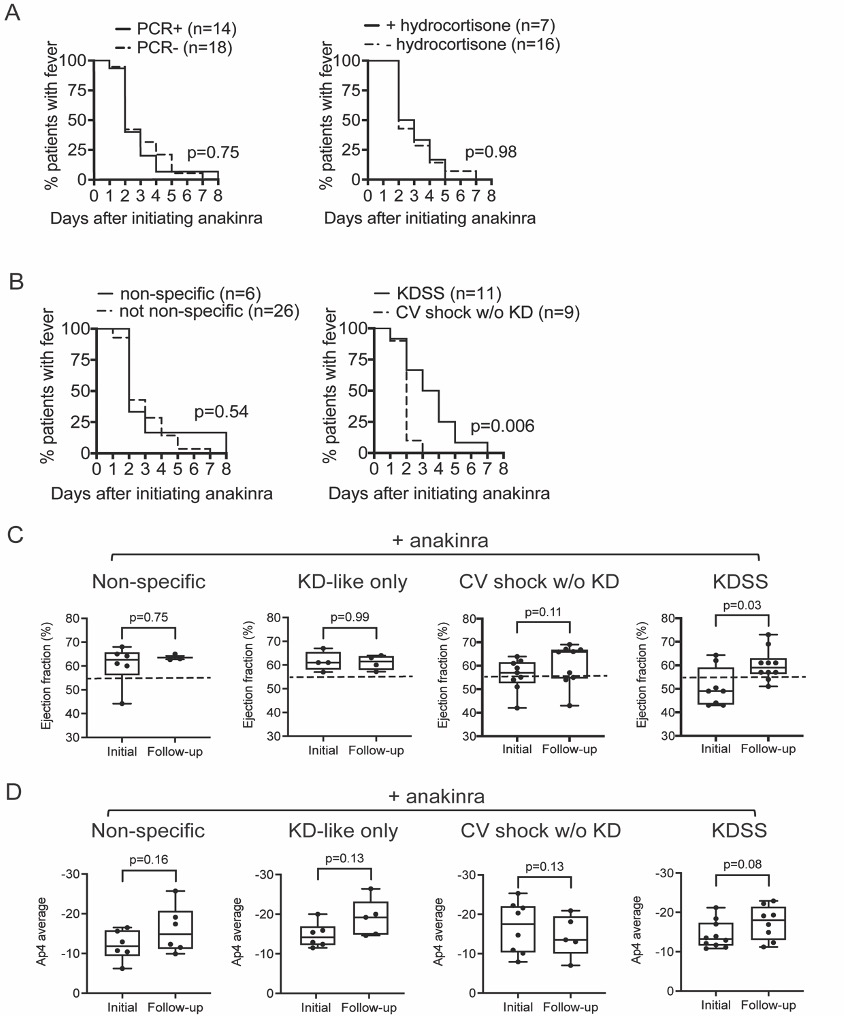 Primary outcome measurement of MIS-C patients with anakinra stratified by non-specific, KD-like only, CV shock only, and KDSS clinical phenotypes. Resolution of fever in MIS-C patients treated with anakinra and stratified by SARS-CoV-2 PCR status or in CV shock patients treated with anakinra and stratified by treatment with stress hydrocortisone are shown in A. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown in C. In D, the effects of anakinra in apical (Ap4) cardiac strain in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown. Data were analyzed for statistical significance by log rank test or non-parametric Wilcoxon matched pairs signed rank test. A p-value < 0.05 was considered statistically significant. KD= Kawasaki disease-like, KDSS= Kawasaki disease shock syndrome, PCR= SAR-CoV2 polymerase chain reaction.
Primary outcome measurement of MIS-C patients with anakinra stratified by non-specific, KD-like only, CV shock only, and KDSS clinical phenotypes. Resolution of fever in MIS-C patients treated with anakinra and stratified by SARS-CoV-2 PCR status or in CV shock patients treated with anakinra and stratified by treatment with stress hydrocortisone are shown in A. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown in C. In D, the effects of anakinra in apical (Ap4) cardiac strain in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown. Data were analyzed for statistical significance by log rank test or non-parametric Wilcoxon matched pairs signed rank test. A p-value < 0.05 was considered statistically significant. KD= Kawasaki disease-like, KDSS= Kawasaki disease shock syndrome, PCR= SAR-CoV2 polymerase chain reaction.
Disclosures: D. Brian, None; C. Redmond, None; E. Gotschlich, None; S. Sule, None; T. Ronis, None; K. Vazzana, None; M. Sherman, None; R. Connor, None; A. Bosk, None; N. Dham, None; A. Harahsheh, OP2 DRUGS (“OP2”); E. Wells, None; R. DeBiasi, None; H. Srinivasalu, None.
Background/Purpose: Multisystem inflammatory syndrome in children (MIS-C) is a clinical entity distinct from primary COVID-19 infection that resembles Kawasaki disease (KD) and toxic shock syndrome. Consensus treatment guidelines for MIS-C have been established based on expert review. Anakinra has been used in refractory cases of MIS-C, but has not been studied in-depth.
Methods: A single center retrospective cohort study was conducted of all MIS-C patients hospitalized at CNH from May 15 to November 15, 2020. Diagnostic and therapeutic data were extracted from the electronic health record. Forty-six patients met the CDC case definition for MIS-C and were further stratified by the presence/absence of KD-like features and CV shock. MIS-C patients with neither KD-like features nor CV shock were termed non-specific, while those with both were termed Kawasaki disease shock syndrome (KDSS).
Results: Of the 46 MIS-C patients included in the study, all received IVIG, 14 (30%) received IVIG monotherapy and 32 (70%) received IVIG+anakinra. No mortality was observed, and no statistically significant differences in adverse reactions were found. The median initial dose of anakinra was 7.9 mg/kg/day to a maximum dose of 9.5 mg/kg/day, with a duration of 9.8 days. Most CV shock patients received anakinra (+anakinra 63% versus -anakinra 21%, p=0.02), which were prescribed at higher doses (+CV shock 9.8 mg/kg/day versus -CV shock 8.0 mg/kg/day, p=0.003) and for a longer duration (+CV shock 10.6 days versus -CV shock 7.1 days, p=0.002). In patients who received IVIG+anakinra, >50% defervesced by day 2 of treatment (figure 1A-C). Among patients receiving IVIG+anakinra, KD-like symptoms was associated with a prolonged time to defervescence (figure 1B). MIS-C patients treated with IVIG+anakinra showed improvement in LVEF (59.0% versus follow-up 62.5%, p=0.004) (figure 1D) and Ap4 cardiac strain (median initial -13.2 versus follow-up -17.8, p=0.04) (figure 1E). While improvements in LVEF with IVIG+anakinra remained significant in CV shock and KDSS subgroup analysis (figure 1D and figure 2C), improvement in cardiac strain was significant in those with KD-like features (median initial -3.2 versus follow-up -19.1, p=0.006) (figure 1D), but not CV shock (p=0.37) (figure 1D and 2D).
Conclusion: Treatment of MIS-C with anakinra was associated with resolution of symptoms, including fever and cardiovascular dysfunction, with no significant adverse events. Our findings suggest an alternative strategy associated with resolution of MIS-C-associated hyperinflammation, and may be useful in MIS-C patients for whom steroids may be unfavorable or contraindicated.
 Primary outcome measurements of MIS-C patients with non-specific, KD-like, and CV shock clinical phenotypes treated with anakinra. The effect of anakinra on defervescence in all patients receiving anakinra (A), as well as those with KD-like features (B) and CV shock phenotypes (C), is shown. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as the LVEF of MIS-C patients with KD-like features or CV shock who received anakinra, is shown in D. The lower limit of normal LVEF (55%) is indicated by the dotted lines. Ap4 cardiac strain on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as MIS-C patients with KD-like features or CV shock who had received anakinra, is shown in E. Data were analyzed for statistical significance by log rank test (B and C) or non-parametric two-tailed Wilcoxon matched pairs signed rank test (D and E), and a p-value < 0.05 was considered statistically significant.
Primary outcome measurements of MIS-C patients with non-specific, KD-like, and CV shock clinical phenotypes treated with anakinra. The effect of anakinra on defervescence in all patients receiving anakinra (A), as well as those with KD-like features (B) and CV shock phenotypes (C), is shown. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as the LVEF of MIS-C patients with KD-like features or CV shock who received anakinra, is shown in D. The lower limit of normal LVEF (55%) is indicated by the dotted lines. Ap4 cardiac strain on initial and repeat echocardiograms in MIS-C patients who did not receive anakinra (-anakinra) with those who received anakinra (+anakinra), as well as MIS-C patients with KD-like features or CV shock who had received anakinra, is shown in E. Data were analyzed for statistical significance by log rank test (B and C) or non-parametric two-tailed Wilcoxon matched pairs signed rank test (D and E), and a p-value < 0.05 was considered statistically significant. Primary outcome measurement of MIS-C patients with anakinra stratified by non-specific, KD-like only, CV shock only, and KDSS clinical phenotypes. Resolution of fever in MIS-C patients treated with anakinra and stratified by SARS-CoV-2 PCR status or in CV shock patients treated with anakinra and stratified by treatment with stress hydrocortisone are shown in A. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown in C. In D, the effects of anakinra in apical (Ap4) cardiac strain in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown. Data were analyzed for statistical significance by log rank test or non-parametric Wilcoxon matched pairs signed rank test. A p-value < 0.05 was considered statistically significant. KD= Kawasaki disease-like, KDSS= Kawasaki disease shock syndrome, PCR= SAR-CoV2 polymerase chain reaction.
Primary outcome measurement of MIS-C patients with anakinra stratified by non-specific, KD-like only, CV shock only, and KDSS clinical phenotypes. Resolution of fever in MIS-C patients treated with anakinra and stratified by SARS-CoV-2 PCR status or in CV shock patients treated with anakinra and stratified by treatment with stress hydrocortisone are shown in A. Left ventricular ejection fractions (LVEF) on initial and repeat echocardiograms in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown in C. In D, the effects of anakinra in apical (Ap4) cardiac strain in MIS-C patients with non-specific, KD-like only, CV shock without KD-like features (w/o KD), and KDSS phenotypes are shown. Data were analyzed for statistical significance by log rank test or non-parametric Wilcoxon matched pairs signed rank test. A p-value < 0.05 was considered statistically significant. KD= Kawasaki disease-like, KDSS= Kawasaki disease shock syndrome, PCR= SAR-CoV2 polymerase chain reaction.Disclosures: D. Brian, None; C. Redmond, None; E. Gotschlich, None; S. Sule, None; T. Ronis, None; K. Vazzana, None; M. Sherman, None; R. Connor, None; A. Bosk, None; N. Dham, None; A. Harahsheh, OP2 DRUGS (“OP2”); E. Wells, None; R. DeBiasi, None; H. Srinivasalu, None.

