Back
Abstract Session
Session: Abstracts: Pediatric Rheumatology – Clinical II: JIA (2209–2214)
2214: Deep Immune Profiling Uncovers Novel Associations with Variable Clinical Phenotypes of Multisystem Inflammatory Syndrome in Children (MIS-C)
Monday, November 14, 2022
4:15 PM – 4:25 PM Eastern Time
Location: Room 119
- CR
Christopher Redmond, MD, MSc
National Institutes of Health
Rockville, MD, United States
Presenting Author(s)
Christopher Redmond1, Moses Kitakule2, Riccardo Castagnoli3, Francesco Licciardi4, Cihan Oguz5, Maria Cecilia Poli6, Aran Son5, Sarah Weber5, Luigi Notarangelo7, Helen Su8 and Daniella Schwartz9, 1National Institutes of Health, Rockville, MD, 2Columbia University, New York, NY, 3National Institute of Allergy and Infectious Diseases, Bethesda, MD, 4OSPEDALE REGINA MARGHERITA, Torino, Italy, 5National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, 6Roberto del Rio, Santiago, Chile, 7NIAID/NIH, Bethesda, MD, 8NIH/NIAID, Bethesda, MD, 9National Institutes of Health Clinical Center, Bethesda, MD
Background/Purpose: Multisystem Inflammatory Syndrome in Children (MIS-C) is a systemic inflammatory condition that follows SARS-CoV2 infection or exposure in children. Clinical presentations are highly variable and include fever, gastrointestinal (GI) disease, shock, and Kawasaki Disease (KD). Compared to patients with acute COVID, patients with MIS-C have a distinct immune signature and expansion of TRVB-11 expressing T cells. However, the relationship between immunological and clinical phenotypes of MIS-C is unknown.
Methods: Serum biomarkers, TCR repertoire, and SARS-CoV2-specific T cell responses were measured in a cohort of 76 MIS-C patients. To quantify SARS-CoV2-specific T cell responses, peripheral blood mononuclear cells (PBMCs) were stimulated with SARS-CoV2 peptides and analyzed by flow cytometry, gated on CD45RO+CD4+ and CD45RO+CD8+ T cells. Mann-Whitney testing with multiple comparison adjustment was used for statistical analysis.
Results: Serum biomarkers associated with macrophage and Th1 activation were elevated in MIS-C patients with shock, consistent with previous reports (Fig 1). Diarrhea was associated with a significant reduction in shock-associated serum biomarkers, suggesting a protective effect. There was a trend towards increased SARS-CoV2-induced IFN-y production in CD45RO+CD4+ cells from patients with shock, and towards reduced IFN-y production in patients with diarrhea (Fig 2). Neurologic symptoms and respiratory failure were associated with significantly increased SARS-CoV2-specific CD4+ T cell responses. TRVB-11 usage was highly associated with KD and coronary aneurysms, suggesting a potential biomarker for these manifestations in MIS-C patients (Fig 2).
Conclusion: By identifying novel immunologic associations with the different clinical phenotypes of MIS-C, this study provides insights into the clinical heterogeneity of MIS-C and suggests potential biomarkers for specific manifestations. Shock and respiratory disease are associated with markers of Th1 and macrophage activation. The inverse association of these markers with diarrhea is consistent with reports that diarrhea is also associated with reduced severity of acute COVID-19. Conversely, persistent fecal SARS-CoV2 viremia can increase serum spike protein levels, which is thought to activate a superantigen pathway that promotes TRVB-11 expansion in MIS-C. The association of TRVB-11 with KD suggests that gastrointestinal SARS-CoV2 involvement may be associated with adverse outcomes. Hence, the different clinical manifestations of MIS-C appear to be associated with unique immunophenotypes that could provide biomarkers for patients at risk for severe disease, like shock or KD.
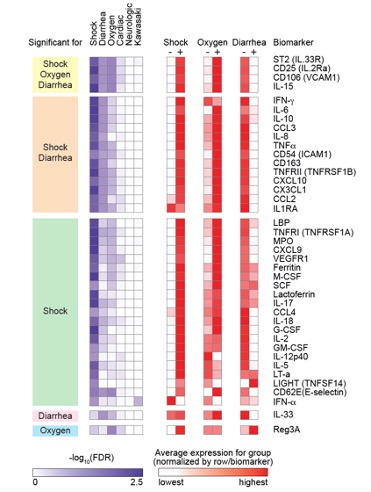 Figure 1 Variation in serum biomarker expression in MIS-C patients based on clinical phenotype
Figure 1 Variation in serum biomarker expression in MIS-C patients based on clinical phenotype
.jpg) Figure 2 PBMCs stimulated with SARS-CoV2 peptides, membrane, spike, nucleocapsid (NC), and all of the peptides pooled together. Cytokine expression was analyzed by flow cytometry in CD4+CD45RO+ T cells. IFNy expression was trended higher in patients with shock (2A) and lower in patients with diarrhea (2B). IFNy expression was significantly higher in patients with neurologic symptoms (2C) and IL2 was significantly higher in patients with respiratory compromise (2D). TRVB-11 usage as significantly higher in MIS-C patients who met the criteria for complete on incomplete Kawasaki Disease and those with documented coronary aneurysms (2E).
Figure 2 PBMCs stimulated with SARS-CoV2 peptides, membrane, spike, nucleocapsid (NC), and all of the peptides pooled together. Cytokine expression was analyzed by flow cytometry in CD4+CD45RO+ T cells. IFNy expression was trended higher in patients with shock (2A) and lower in patients with diarrhea (2B). IFNy expression was significantly higher in patients with neurologic symptoms (2C) and IL2 was significantly higher in patients with respiratory compromise (2D). TRVB-11 usage as significantly higher in MIS-C patients who met the criteria for complete on incomplete Kawasaki Disease and those with documented coronary aneurysms (2E).
Disclosures: C. Redmond, None; M. Kitakule, None; R. Castagnoli, None; F. Licciardi, None; C. Oguz, None; M. Poli, None; A. Son, None; S. Weber, None; L. Notarangelo, None; H. Su, Amgen, Pfizer, Eli Lilly, Avanos Medical, Procter & Gamble, Viatris, Zimmer Biomet; D. Schwartz, None.
Background/Purpose: Multisystem Inflammatory Syndrome in Children (MIS-C) is a systemic inflammatory condition that follows SARS-CoV2 infection or exposure in children. Clinical presentations are highly variable and include fever, gastrointestinal (GI) disease, shock, and Kawasaki Disease (KD). Compared to patients with acute COVID, patients with MIS-C have a distinct immune signature and expansion of TRVB-11 expressing T cells. However, the relationship between immunological and clinical phenotypes of MIS-C is unknown.
Methods: Serum biomarkers, TCR repertoire, and SARS-CoV2-specific T cell responses were measured in a cohort of 76 MIS-C patients. To quantify SARS-CoV2-specific T cell responses, peripheral blood mononuclear cells (PBMCs) were stimulated with SARS-CoV2 peptides and analyzed by flow cytometry, gated on CD45RO+CD4+ and CD45RO+CD8+ T cells. Mann-Whitney testing with multiple comparison adjustment was used for statistical analysis.
Results: Serum biomarkers associated with macrophage and Th1 activation were elevated in MIS-C patients with shock, consistent with previous reports (Fig 1). Diarrhea was associated with a significant reduction in shock-associated serum biomarkers, suggesting a protective effect. There was a trend towards increased SARS-CoV2-induced IFN-y production in CD45RO+CD4+ cells from patients with shock, and towards reduced IFN-y production in patients with diarrhea (Fig 2). Neurologic symptoms and respiratory failure were associated with significantly increased SARS-CoV2-specific CD4+ T cell responses. TRVB-11 usage was highly associated with KD and coronary aneurysms, suggesting a potential biomarker for these manifestations in MIS-C patients (Fig 2).
Conclusion: By identifying novel immunologic associations with the different clinical phenotypes of MIS-C, this study provides insights into the clinical heterogeneity of MIS-C and suggests potential biomarkers for specific manifestations. Shock and respiratory disease are associated with markers of Th1 and macrophage activation. The inverse association of these markers with diarrhea is consistent with reports that diarrhea is also associated with reduced severity of acute COVID-19. Conversely, persistent fecal SARS-CoV2 viremia can increase serum spike protein levels, which is thought to activate a superantigen pathway that promotes TRVB-11 expansion in MIS-C. The association of TRVB-11 with KD suggests that gastrointestinal SARS-CoV2 involvement may be associated with adverse outcomes. Hence, the different clinical manifestations of MIS-C appear to be associated with unique immunophenotypes that could provide biomarkers for patients at risk for severe disease, like shock or KD.
 Figure 1 Variation in serum biomarker expression in MIS-C patients based on clinical phenotype
Figure 1 Variation in serum biomarker expression in MIS-C patients based on clinical phenotype.jpg) Figure 2 PBMCs stimulated with SARS-CoV2 peptides, membrane, spike, nucleocapsid (NC), and all of the peptides pooled together. Cytokine expression was analyzed by flow cytometry in CD4+CD45RO+ T cells. IFNy expression was trended higher in patients with shock (2A) and lower in patients with diarrhea (2B). IFNy expression was significantly higher in patients with neurologic symptoms (2C) and IL2 was significantly higher in patients with respiratory compromise (2D). TRVB-11 usage as significantly higher in MIS-C patients who met the criteria for complete on incomplete Kawasaki Disease and those with documented coronary aneurysms (2E).
Figure 2 PBMCs stimulated with SARS-CoV2 peptides, membrane, spike, nucleocapsid (NC), and all of the peptides pooled together. Cytokine expression was analyzed by flow cytometry in CD4+CD45RO+ T cells. IFNy expression was trended higher in patients with shock (2A) and lower in patients with diarrhea (2B). IFNy expression was significantly higher in patients with neurologic symptoms (2C) and IL2 was significantly higher in patients with respiratory compromise (2D). TRVB-11 usage as significantly higher in MIS-C patients who met the criteria for complete on incomplete Kawasaki Disease and those with documented coronary aneurysms (2E). Disclosures: C. Redmond, None; M. Kitakule, None; R. Castagnoli, None; F. Licciardi, None; C. Oguz, None; M. Poli, None; A. Son, None; S. Weber, None; L. Notarangelo, None; H. Su, Amgen, Pfizer, Eli Lilly, Avanos Medical, Procter & Gamble, Viatris, Zimmer Biomet; D. Schwartz, None.

