Back
Abstract Session
Session: Abstracts: SLE – Etiology and Pathogenesis (2264–2267)
2267: Plasmacytoid Dendritic Cells Are Not Major Producers of Type 1 Interferons in Cutaneous Lupus
Monday, November 14, 2022
5:45 PM – 5:55 PM Eastern Time
Location: Room 113
- TV
Thomas Vazquez, MD
University of Pennsylvania
Philadelphia, PA, United States
Presenting Author(s)
Thomas Vazquez1, Nilesh Kodali1, DeAnna Diaz2, Jay Patel3, Emily Keyes2, Grant Sprow2, Meena Sharma1, Mariko Ogawa-Momohara2, Madison Grinnell2, Josh Dan2 and Victoria Werth2, 1Philadelphia VAMC, Philadelphia, PA, USA and Department of Dermatology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, PA, 2Philadelphia VAMC, Philadelphia, PA, USA and Department of Dermatology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, 3Philadelphia VAMC, Philadelphia, PA, USA and Department of Dermatology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, PN
Background/Purpose: Type 1 interferons (IFN-1) are major drivers of disease activity in systemic (SLE) and cutaneous lupus erythematosus (CLE). Plasmacytoid dendritic cells (pDCs) are the major producers of IFN-1 during viral infection. Therefore, pDCs have been hypothesized to be the primary IFN-1 producing cell in lupus. IFN-1 production by pDCs has not been extensively studied due to reliance on interferon gene signatures as a proxy for IFN-1 levels as well as a reliance on pDCs from healthy controls for in vitro studies. However, it has been known that pDCs are reduced in the circulation in SLE and have also been more recently shown to have an exhausted phenotype, suggesting that pathogenic pDCs may be located in the end organs.
Methods: Well characterized CLE patients were recruited from a prospective database. We performed imaging mass cytometry on archived treatment naïve CLE skin (n=48) and a subset were also used for mRNA in situ hybridization. Fresh 4mm punch biopsies (n=3) and blood (n=4) were obtained from CLE patients and healthy controls (n=3) for multiplexed flow cytometry or cytometry by time of flight (CyTOF).
Results: In lesional CLE skin, only 8.5% (IQR 0.0-22.22) of pDCs were IFNa+. Across all CLE biopsies, pDCs were the second lowest contributor of absolute IFNα+ cells (Median 1; IQR 0-4.5), with B lymphocytes being the smallest contributor. Classic dendritic cells (cDCs) and macrophages (Mφ) were the largest relative and absolute contributors of IFNα in CLE. For IFNβ, only 16.7% of pDCs were positive, compared to 60.7% of CD14+CD16+ Mφ. pDCs were also the second lowest contributors of IFNβ+ cells (Median 1; IQR 0-7), with B lymphocytes again being the lowest. IFN-1 mRNA was detected in ITGAX (CD11c) positive cDCs but we did not identify colocalization of pDC-specific gene, CLEC4C (BDCA2), and IFN-1 mRNA. Flow cytometry on leukocytes eluted from fresh lesional biopsies showed a median of 5.2% of pDCs were positive for IFNα (IQR 3.85 – 9.64) which was significantly lower than the median of 37.9% of HC PBMC pDCs (34.5 – 39.8) (p< 0.0001). In comparison, other leukocytes in CLE skin displayed greater positivity for IFNα such as CD4 T cells (Median, IQR) (24.5%, 10.2 – 29), CD14+ cells (17.6%, 10.7 – 23.1), Classical Monocytes (CD14++CD16-) (11.4%, 10.2 – 20.2), Intermediate Monocytes (CD14++CD16+) (13.7%, 12.4 – 17.5), CD68+ Macrophages (24.6%, 10.9 – 32.4), M1 Macrophages (CD68+CD163-) (23.7%, 21.6 – 31.7), M2 Macrophages (CD68+CD163+) (35.9%, 26.4 – 40.7), and CD16+ cells (25.7%, 20.4 – 29.5). Further verification using a Z-axis overlay of intracellular markers on tSNE plots of immune cell clusters identified by CyTOF confirmed low expression of IFN-1and the interferogenic pathway, phosphorylated stimulator of interferon genes (pSTING), in the pDCs.
Conclusion: Taken together, these findings suggest pDCs may not play the central role in CLE as major IFN-1 producers and myeloid cells are larger contributors of IFN-1 in numbers and as a percent. pDCs may have a pathogenic role in CLE through IFN-1-independent mechanisms.
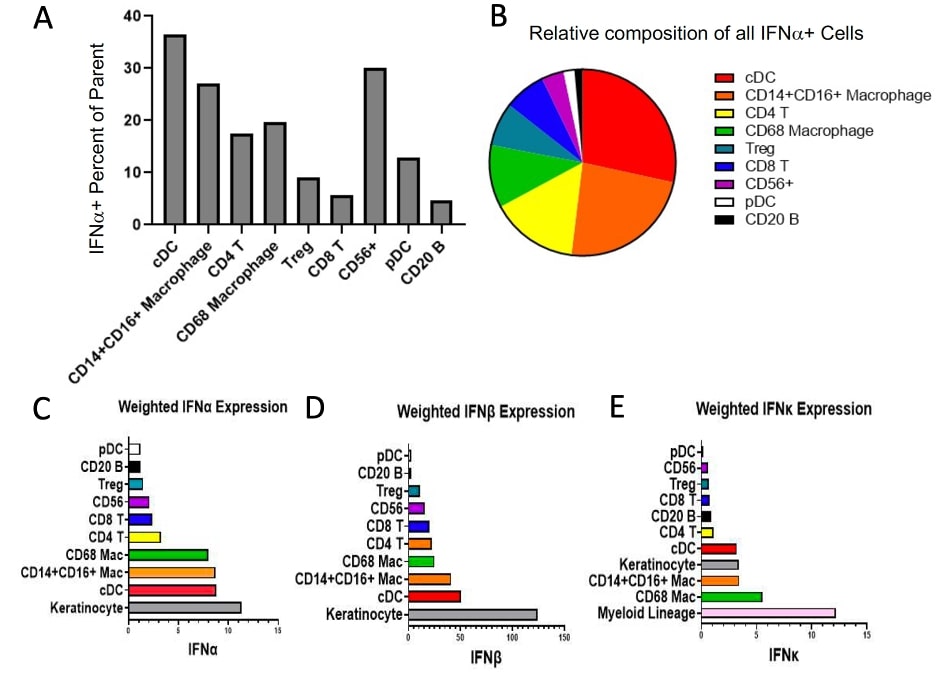 A) IFNα+ Percent of Parent for cell types identified in CLE skin shown in order of descending absolute contribution of IFNα+ cell counts in CLE. 12.83% of pDCs are positive for IFNα. B) Composition of IFNα+ cells (absolute counts) across all aggregated CLE biopsies. C) Weighted (cell count x MPI) IFNα expression. D) Weighted IFNβ expression. E) Weighted IFNκ expression with myeloid lineage consisting of CD68 Mac, CD14+CD16+ Mac, and cDCs revealing dominant IFN-1 contribution from myeloid cells followed by keratinocytes.
A) IFNα+ Percent of Parent for cell types identified in CLE skin shown in order of descending absolute contribution of IFNα+ cell counts in CLE. 12.83% of pDCs are positive for IFNα. B) Composition of IFNα+ cells (absolute counts) across all aggregated CLE biopsies. C) Weighted (cell count x MPI) IFNα expression. D) Weighted IFNβ expression. E) Weighted IFNκ expression with myeloid lineage consisting of CD68 Mac, CD14+CD16+ Mac, and cDCs revealing dominant IFN-1 contribution from myeloid cells followed by keratinocytes.
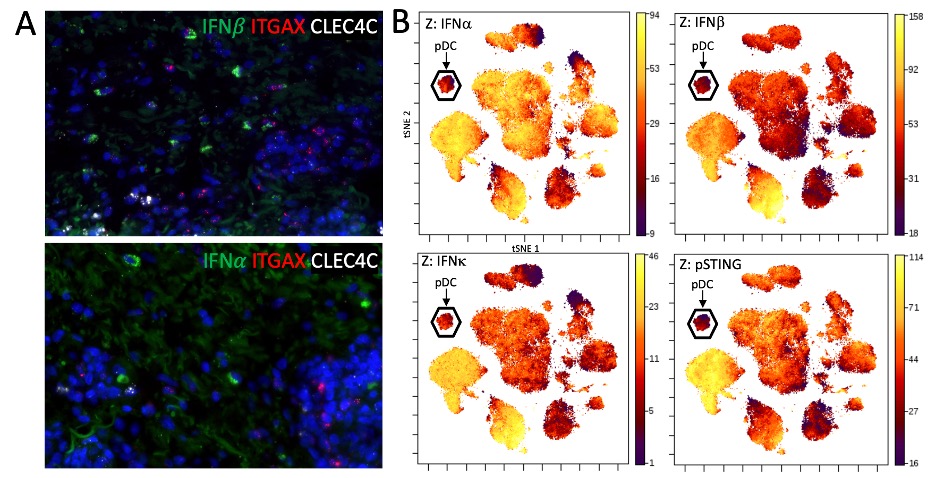 A) Representative images of mRNA in situ hybridization of IFN and IFN mRNA with markers for conventional dendritic cells (ITGAX) and plasmacytoid dendritic cells (CLEC4C). No observable overlap of type 1 interferon mRNA and CLEC4C was seen. B) tSNE plot of PBMCs from a CLE patient stained and acquired by CyTOF. Plasmacytoid dendritic cells, identified principally on CD123 expression, demonstrate little type 1 interferon staining as well as activation of the STING pathway (a signaling molecule upstream of type 1 interferon transcription) on the Z axis.
A) Representative images of mRNA in situ hybridization of IFN and IFN mRNA with markers for conventional dendritic cells (ITGAX) and plasmacytoid dendritic cells (CLEC4C). No observable overlap of type 1 interferon mRNA and CLEC4C was seen. B) tSNE plot of PBMCs from a CLE patient stained and acquired by CyTOF. Plasmacytoid dendritic cells, identified principally on CD123 expression, demonstrate little type 1 interferon staining as well as activation of the STING pathway (a signaling molecule upstream of type 1 interferon transcription) on the Z axis.
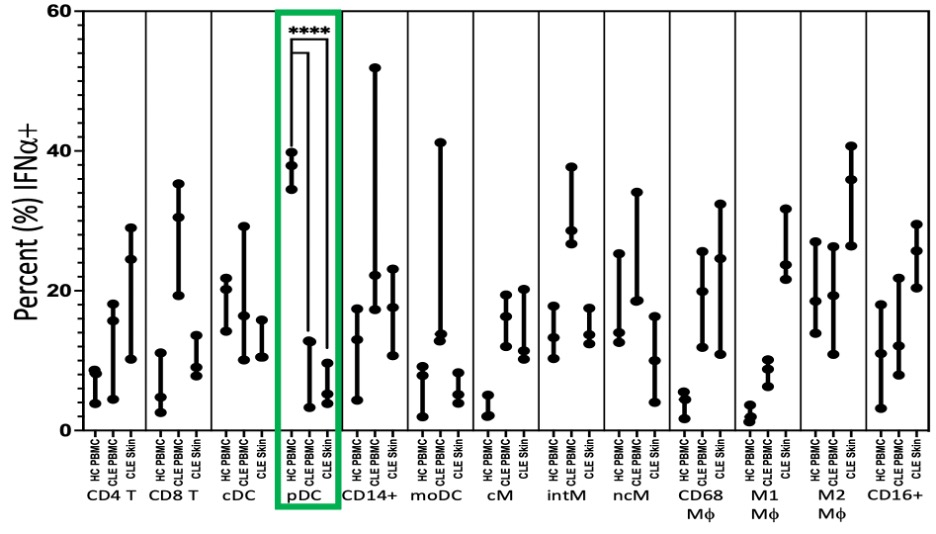 Flow cytometry of leukocytes eluted from CLE skin demonstrating significantly fewer IFN+ plasmacytoid dendritic cells (pDCs) in cutaneous lupus erythematosus (CLE) skin (Median 5.2%, p < 0.0001) and peripheral blood mononuclear cells (PBMCs) (12.7%, p < 0.0001) than healthy control PBMCs (37.9%). Abbreviations: cDC: classical dendritic cell; pDC: plasmacytoid dendritic cell; moDC: monocyte derived dendritic cell; cM: classical monocyte; intM: intermediate monocyte; ncM: nonclassical monocyte; Mf: macrophage.
Flow cytometry of leukocytes eluted from CLE skin demonstrating significantly fewer IFN+ plasmacytoid dendritic cells (pDCs) in cutaneous lupus erythematosus (CLE) skin (Median 5.2%, p < 0.0001) and peripheral blood mononuclear cells (PBMCs) (12.7%, p < 0.0001) than healthy control PBMCs (37.9%). Abbreviations: cDC: classical dendritic cell; pDC: plasmacytoid dendritic cell; moDC: monocyte derived dendritic cell; cM: classical monocyte; intM: intermediate monocyte; ncM: nonclassical monocyte; Mf: macrophage.
Disclosures: T. Vazquez, None; N. Kodali, None; D. Diaz, None; J. Patel, None; E. Keyes, None; G. Sprow, None; M. Sharma, None; M. Ogawa-Momohara, None; M. Grinnell, None; J. Dan, None; V. Werth, GlaxoSmithKline, CLASI.
Background/Purpose: Type 1 interferons (IFN-1) are major drivers of disease activity in systemic (SLE) and cutaneous lupus erythematosus (CLE). Plasmacytoid dendritic cells (pDCs) are the major producers of IFN-1 during viral infection. Therefore, pDCs have been hypothesized to be the primary IFN-1 producing cell in lupus. IFN-1 production by pDCs has not been extensively studied due to reliance on interferon gene signatures as a proxy for IFN-1 levels as well as a reliance on pDCs from healthy controls for in vitro studies. However, it has been known that pDCs are reduced in the circulation in SLE and have also been more recently shown to have an exhausted phenotype, suggesting that pathogenic pDCs may be located in the end organs.
Methods: Well characterized CLE patients were recruited from a prospective database. We performed imaging mass cytometry on archived treatment naïve CLE skin (n=48) and a subset were also used for mRNA in situ hybridization. Fresh 4mm punch biopsies (n=3) and blood (n=4) were obtained from CLE patients and healthy controls (n=3) for multiplexed flow cytometry or cytometry by time of flight (CyTOF).
Results: In lesional CLE skin, only 8.5% (IQR 0.0-22.22) of pDCs were IFNa+. Across all CLE biopsies, pDCs were the second lowest contributor of absolute IFNα+ cells (Median 1; IQR 0-4.5), with B lymphocytes being the smallest contributor. Classic dendritic cells (cDCs) and macrophages (Mφ) were the largest relative and absolute contributors of IFNα in CLE. For IFNβ, only 16.7% of pDCs were positive, compared to 60.7% of CD14+CD16+ Mφ. pDCs were also the second lowest contributors of IFNβ+ cells (Median 1; IQR 0-7), with B lymphocytes again being the lowest. IFN-1 mRNA was detected in ITGAX (CD11c) positive cDCs but we did not identify colocalization of pDC-specific gene, CLEC4C (BDCA2), and IFN-1 mRNA. Flow cytometry on leukocytes eluted from fresh lesional biopsies showed a median of 5.2% of pDCs were positive for IFNα (IQR 3.85 – 9.64) which was significantly lower than the median of 37.9% of HC PBMC pDCs (34.5 – 39.8) (p< 0.0001). In comparison, other leukocytes in CLE skin displayed greater positivity for IFNα such as CD4 T cells (Median, IQR) (24.5%, 10.2 – 29), CD14+ cells (17.6%, 10.7 – 23.1), Classical Monocytes (CD14++CD16-) (11.4%, 10.2 – 20.2), Intermediate Monocytes (CD14++CD16+) (13.7%, 12.4 – 17.5), CD68+ Macrophages (24.6%, 10.9 – 32.4), M1 Macrophages (CD68+CD163-) (23.7%, 21.6 – 31.7), M2 Macrophages (CD68+CD163+) (35.9%, 26.4 – 40.7), and CD16+ cells (25.7%, 20.4 – 29.5). Further verification using a Z-axis overlay of intracellular markers on tSNE plots of immune cell clusters identified by CyTOF confirmed low expression of IFN-1and the interferogenic pathway, phosphorylated stimulator of interferon genes (pSTING), in the pDCs.
Conclusion: Taken together, these findings suggest pDCs may not play the central role in CLE as major IFN-1 producers and myeloid cells are larger contributors of IFN-1 in numbers and as a percent. pDCs may have a pathogenic role in CLE through IFN-1-independent mechanisms.
 A) IFNα+ Percent of Parent for cell types identified in CLE skin shown in order of descending absolute contribution of IFNα+ cell counts in CLE. 12.83% of pDCs are positive for IFNα. B) Composition of IFNα+ cells (absolute counts) across all aggregated CLE biopsies. C) Weighted (cell count x MPI) IFNα expression. D) Weighted IFNβ expression. E) Weighted IFNκ expression with myeloid lineage consisting of CD68 Mac, CD14+CD16+ Mac, and cDCs revealing dominant IFN-1 contribution from myeloid cells followed by keratinocytes.
A) IFNα+ Percent of Parent for cell types identified in CLE skin shown in order of descending absolute contribution of IFNα+ cell counts in CLE. 12.83% of pDCs are positive for IFNα. B) Composition of IFNα+ cells (absolute counts) across all aggregated CLE biopsies. C) Weighted (cell count x MPI) IFNα expression. D) Weighted IFNβ expression. E) Weighted IFNκ expression with myeloid lineage consisting of CD68 Mac, CD14+CD16+ Mac, and cDCs revealing dominant IFN-1 contribution from myeloid cells followed by keratinocytes.  A) Representative images of mRNA in situ hybridization of IFN and IFN mRNA with markers for conventional dendritic cells (ITGAX) and plasmacytoid dendritic cells (CLEC4C). No observable overlap of type 1 interferon mRNA and CLEC4C was seen. B) tSNE plot of PBMCs from a CLE patient stained and acquired by CyTOF. Plasmacytoid dendritic cells, identified principally on CD123 expression, demonstrate little type 1 interferon staining as well as activation of the STING pathway (a signaling molecule upstream of type 1 interferon transcription) on the Z axis.
A) Representative images of mRNA in situ hybridization of IFN and IFN mRNA with markers for conventional dendritic cells (ITGAX) and plasmacytoid dendritic cells (CLEC4C). No observable overlap of type 1 interferon mRNA and CLEC4C was seen. B) tSNE plot of PBMCs from a CLE patient stained and acquired by CyTOF. Plasmacytoid dendritic cells, identified principally on CD123 expression, demonstrate little type 1 interferon staining as well as activation of the STING pathway (a signaling molecule upstream of type 1 interferon transcription) on the Z axis.  Flow cytometry of leukocytes eluted from CLE skin demonstrating significantly fewer IFN+ plasmacytoid dendritic cells (pDCs) in cutaneous lupus erythematosus (CLE) skin (Median 5.2%, p < 0.0001) and peripheral blood mononuclear cells (PBMCs) (12.7%, p < 0.0001) than healthy control PBMCs (37.9%). Abbreviations: cDC: classical dendritic cell; pDC: plasmacytoid dendritic cell; moDC: monocyte derived dendritic cell; cM: classical monocyte; intM: intermediate monocyte; ncM: nonclassical monocyte; Mf: macrophage.
Flow cytometry of leukocytes eluted from CLE skin demonstrating significantly fewer IFN+ plasmacytoid dendritic cells (pDCs) in cutaneous lupus erythematosus (CLE) skin (Median 5.2%, p < 0.0001) and peripheral blood mononuclear cells (PBMCs) (12.7%, p < 0.0001) than healthy control PBMCs (37.9%). Abbreviations: cDC: classical dendritic cell; pDC: plasmacytoid dendritic cell; moDC: monocyte derived dendritic cell; cM: classical monocyte; intM: intermediate monocyte; ncM: nonclassical monocyte; Mf: macrophage.Disclosures: T. Vazquez, None; N. Kodali, None; D. Diaz, None; J. Patel, None; E. Keyes, None; G. Sprow, None; M. Sharma, None; M. Ogawa-Momohara, None; M. Grinnell, None; J. Dan, None; V. Werth, GlaxoSmithKline, CLASI.

