Back
Poster Session A
Rheumatoid arthritis (RA)
Session: (0241–0271) RA – Diagnosis, Manifestations, and Outcomes Poster I
0256: Patients with ACPA-positive and ACPA-negative Rheumatoid Arthritis Show Different Circulating Auto-antibody Repertoires
Saturday, November 12, 2022
1:00 PM – 3:00 PM Eastern Time
Location: Virtual Poster Hall
- KC
Kevin Cunningham, PhD
University of Minnesota
Minneapolis, MN, United States
Abstract Poster Presenter(s)
Kevin Cunningham1, Benjamin Hur2, John Davis2 and Jaeyun Sung2, 1University of Minnesota, Rochester, MN, 2Mayo Clinic, Rochester, MN
Background/Purpose: RA is commonly diagnosed through a serological test for the presence of ACPA, and RA patients who test positive are collectively known as 'ACPA-positive RA' (ACPA+). However, patients can test negative for ACPA yet still be clinically diagnosed with RA, and are thereby designated as 'ACPA-negative RA' (ACPA–). Although ACPA+ and ACPA– patients are known to have different rates of erosive damage and distinct risk factors, the differences in serological profiles between these two disease subgroups are largely unknown. In this study, we performed a comprehensive survey of serum autoantibodies in patients with ACPA+ and ACPA– RA to obtain novel insights into autoantigens underlying the distinct subgroups of RA.
Methods: Serum was collected from patients with ACPA+ RA (n = 32), patients with ACPA– RA (n = 30), and healthy controls (n = 30) (Fig. 1A). Sengenics KREXTM multiplexed autoantibody-screening protein microarray was used to screen for 1,622 different autoantibodies from each serum sample. A Mann–Whitney U test combined with Cliff's delta was used to identify differentially abundant autoantibodies.
Results: ACPA+ RA patients, ACPA– RA patients, and healthy controls displayed differences in their serum autoantibody profiles (Fig. 1B–D). Upon closer examination, we identified differential serum autoantibodies of ACPA+ and ACPA– RA. Specifically, we found 22 and 19 autoantibodies higher in ACPA+ RA patients and ACPA– RA patients, respectively, compared to healthy controls (Fig. 2A–B). Notably, only one autoantibody for GTF2A2 was found to be higher in both RA subgroups. Next, we found autoantibodies for the CISH protein to be positively correlated with the Clinical Disease Activity Index (ρ = 0.44, P = 6.5 × 10-4) (Fig. 3). Additionally, we determined the functional classes of the proteins targeted by the differentially abundant autoantibodies. We found eight different functional classes, using the PANTHER classification system, for the autoantibodies higher in ACPA+ RA patients, and seven different functional classes for the autoantibodies higher in ACPA– RA patients. When comparing these two groups of functional classes, we identified four in common: 'Gene-specific Transcriptional Regulator', 'Nucleic Acid Metabolism Protein', 'Metabolite Interconversion Enzyme', and 'Protein-modifying Enzyme'. This suggests that an imbalance in autoantibody production targeting metabolic and transcription processes could potentially be a newly identified hallmark of RA.
Conclusion: We identified several differentially abundant autoantibodies in ACPA+ and ACPA– RA, many of which were identified for the first time. Our study demonstrates the potential utility of serum autoantibodies as diagnostic targets for RA subgroups and reveals potential therapeutic treatments.
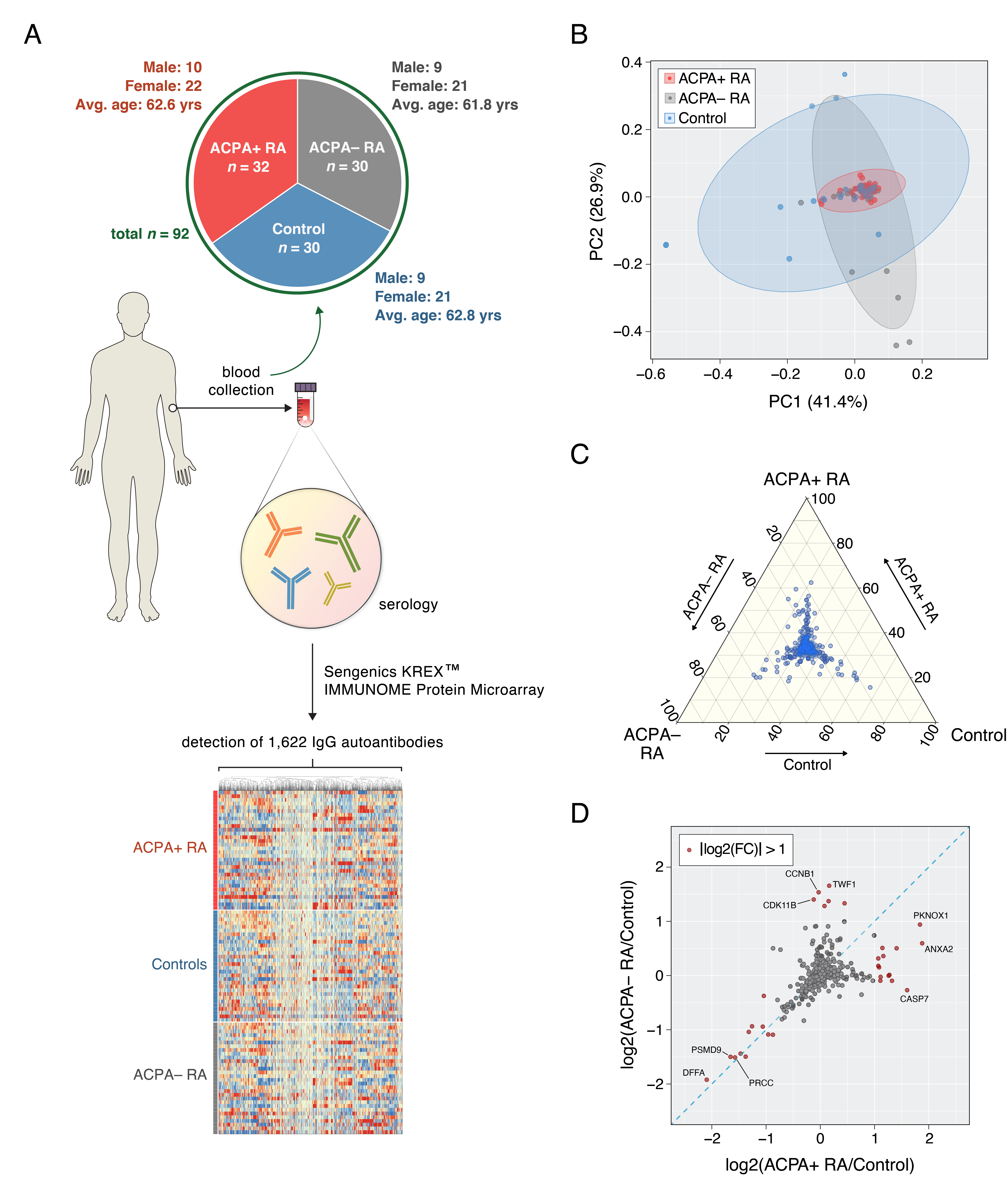 Figure 1. Study design overview and group-wise comparisons of autoantibody composition profiles. (A) Blood (serum) samples were collected to investigate serum autoantibody compositions in ACPA+ RA (n = 32), ACPA– RA (n = 30), and healthy controls (n = 30). Each serum sample was screened for 1,622 IgG autoantibodies using the Sengenics KREXTM IMMUNOME Protein Microarray. Raw relative fluorescence units (RFU) values representing serum autoantibody abundances were transformed using quantile normalization prior to all downstream analyses. Heatmap illustrates autoantibodies clustered according to abundance similarities across samples. (B) Principal component analysis on the 92 autoantibody profiles from ACPA+, ACPA–, and healthy controls. Controls showed the highest within-group heterogeneity in autoantibody composition whereas the ACPA+ RA group showed the least within-group heterogeneity. (C) Ternary plot showing mean abundances of 1,622 autoantibodies across ACPA+, ACPA–, and healthy controls. Coordinates of each point correspond to percentages of mean abundances and sum to 100. Despite most autoantibodies having similar mean abundances across the three study groups, several autoantibodies were found to have high abundance in one group (see points closer to the corners). (D) Fold-changes in mean abundances of 1,622 autoantibodies for the RA subgroups compared to the control group. x-axis and y-axis correspond to the fold-changes between ACPA+ RA and controls and between ACPA– RA and controls, respectively. Autoantibodies shown in red points have a fold-change of 2 (or greater) between groups. As observed in (C), several autoantibodies show fold-changes specific to an RA subgroup. The blue diagonal dashed line represents the line y=x.
Figure 1. Study design overview and group-wise comparisons of autoantibody composition profiles. (A) Blood (serum) samples were collected to investigate serum autoantibody compositions in ACPA+ RA (n = 32), ACPA– RA (n = 30), and healthy controls (n = 30). Each serum sample was screened for 1,622 IgG autoantibodies using the Sengenics KREXTM IMMUNOME Protein Microarray. Raw relative fluorescence units (RFU) values representing serum autoantibody abundances were transformed using quantile normalization prior to all downstream analyses. Heatmap illustrates autoantibodies clustered according to abundance similarities across samples. (B) Principal component analysis on the 92 autoantibody profiles from ACPA+, ACPA–, and healthy controls. Controls showed the highest within-group heterogeneity in autoantibody composition whereas the ACPA+ RA group showed the least within-group heterogeneity. (C) Ternary plot showing mean abundances of 1,622 autoantibodies across ACPA+, ACPA–, and healthy controls. Coordinates of each point correspond to percentages of mean abundances and sum to 100. Despite most autoantibodies having similar mean abundances across the three study groups, several autoantibodies were found to have high abundance in one group (see points closer to the corners). (D) Fold-changes in mean abundances of 1,622 autoantibodies for the RA subgroups compared to the control group. x-axis and y-axis correspond to the fold-changes between ACPA+ RA and controls and between ACPA– RA and controls, respectively. Autoantibodies shown in red points have a fold-change of 2 (or greater) between groups. As observed in (C), several autoantibodies show fold-changes specific to an RA subgroup. The blue diagonal dashed line represents the line y=x.
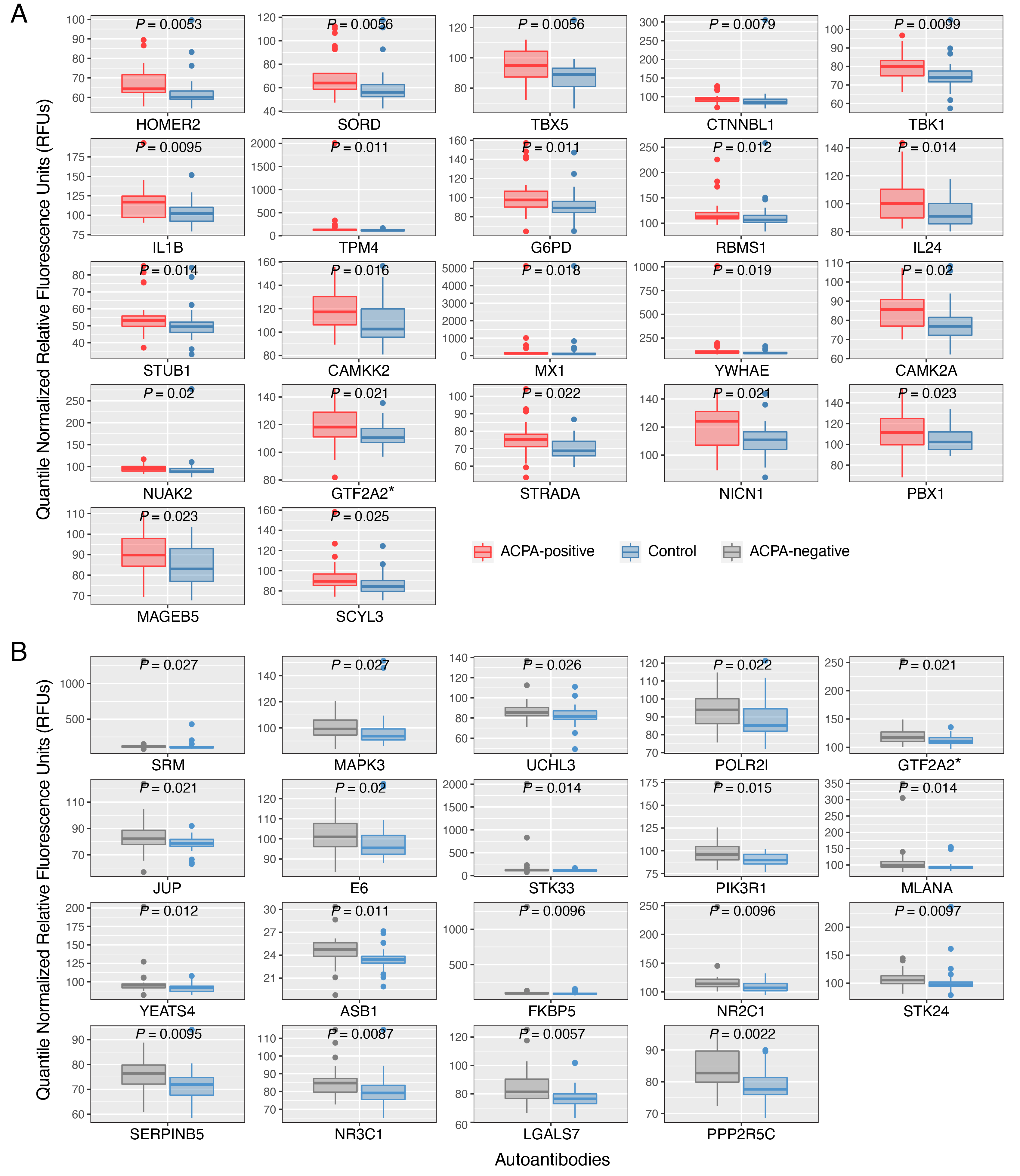 Figure 2. Autoantibodies identified to have significantly higher abundances in two subgroups of rheumatoid arthritis (ACPA+ RA and ACPA– RA) than in healthy controls. Patients with (A) ACPA+ RA and (B) ACPA– RA show higher abundances in 22 and 19 autoantibodies, respectively. Two-sided Mann–Whitney U test (P < 0.05) and Cliff’s Delta (|d| > 0.33) were used to find autoantibodies of significantly higher abundance. Standard box-and-whisker plots (e.g., center line, median; box limits, upper and lower quartiles; whiskers, 1.5× interquartile range; points, outliers) are used to show numerical data. *, auto-antibody for GTF2A2 protein was found to have significantly higher abundances in both ACPA+ and ACPA– RA patient groups than in healthy controls.
Figure 2. Autoantibodies identified to have significantly higher abundances in two subgroups of rheumatoid arthritis (ACPA+ RA and ACPA– RA) than in healthy controls. Patients with (A) ACPA+ RA and (B) ACPA– RA show higher abundances in 22 and 19 autoantibodies, respectively. Two-sided Mann–Whitney U test (P < 0.05) and Cliff’s Delta (|d| > 0.33) were used to find autoantibodies of significantly higher abundance. Standard box-and-whisker plots (e.g., center line, median; box limits, upper and lower quartiles; whiskers, 1.5× interquartile range; points, outliers) are used to show numerical data. *, auto-antibody for GTF2A2 protein was found to have significantly higher abundances in both ACPA+ and ACPA– RA patient groups than in healthy controls.
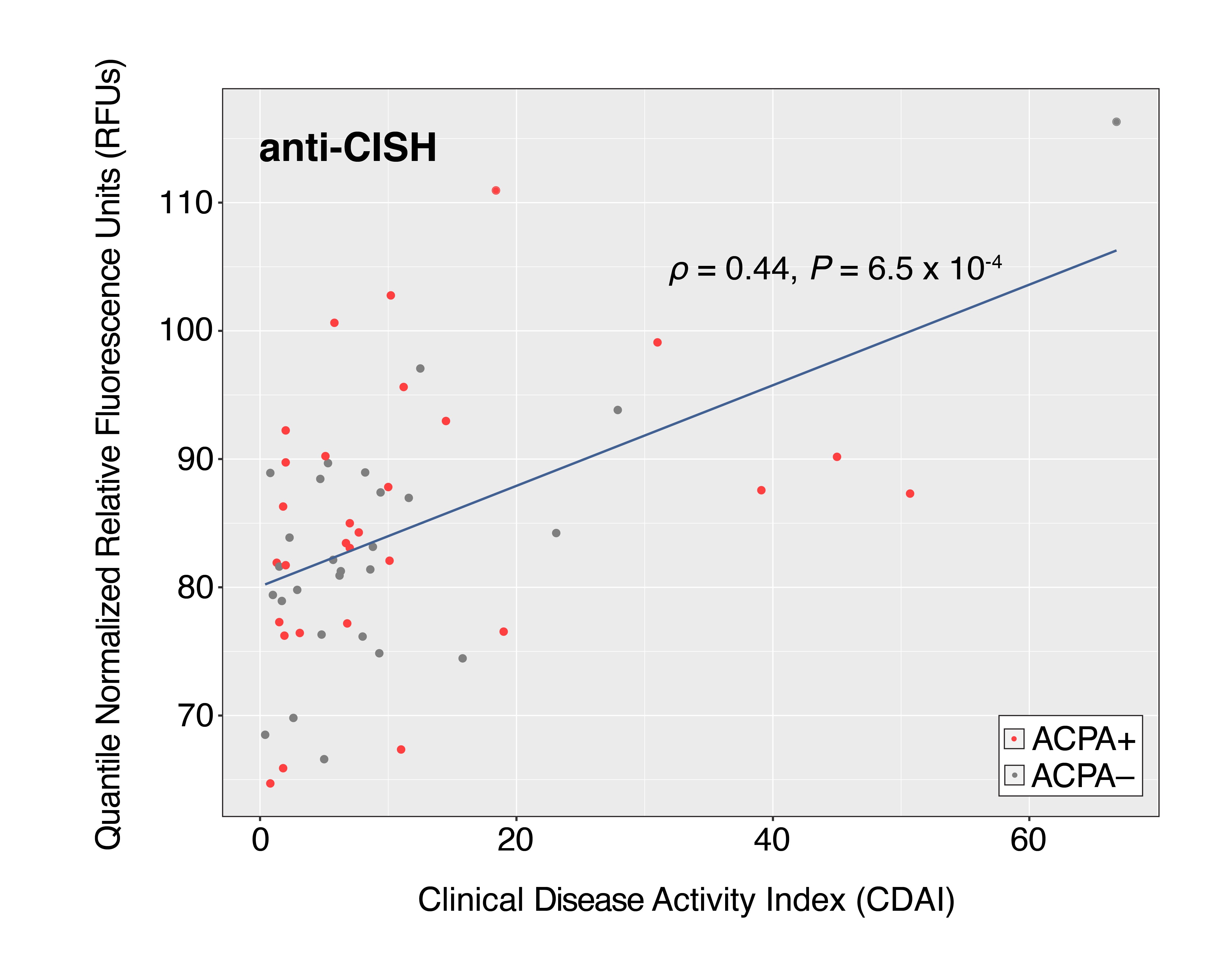 Figure 3. Autoantibodies targeting CISH proteins display the highest correlation with Clinical Disease Activity Index (CDAI). The blue line in the scatterplots represents the linear regression fit of Y~X, wherein X and Y correspond to CDAI and autoantibody abundance, respectively. The Spearman correlation coefficient ρ was used to measure the strength of the relationship between the two variables. Scatterplot contains 56 points corresponding to the number of RA patients whose CDAI scores were available.
Figure 3. Autoantibodies targeting CISH proteins display the highest correlation with Clinical Disease Activity Index (CDAI). The blue line in the scatterplots represents the linear regression fit of Y~X, wherein X and Y correspond to CDAI and autoantibody abundance, respectively. The Spearman correlation coefficient ρ was used to measure the strength of the relationship between the two variables. Scatterplot contains 56 points corresponding to the number of RA patients whose CDAI scores were available.
Disclosures: K. Cunningham, None; B. Hur, None; J. Davis, Pfizer; J. Sung, None.
Background/Purpose: RA is commonly diagnosed through a serological test for the presence of ACPA, and RA patients who test positive are collectively known as 'ACPA-positive RA' (ACPA+). However, patients can test negative for ACPA yet still be clinically diagnosed with RA, and are thereby designated as 'ACPA-negative RA' (ACPA–). Although ACPA+ and ACPA– patients are known to have different rates of erosive damage and distinct risk factors, the differences in serological profiles between these two disease subgroups are largely unknown. In this study, we performed a comprehensive survey of serum autoantibodies in patients with ACPA+ and ACPA– RA to obtain novel insights into autoantigens underlying the distinct subgroups of RA.
Methods: Serum was collected from patients with ACPA+ RA (n = 32), patients with ACPA– RA (n = 30), and healthy controls (n = 30) (Fig. 1A). Sengenics KREXTM multiplexed autoantibody-screening protein microarray was used to screen for 1,622 different autoantibodies from each serum sample. A Mann–Whitney U test combined with Cliff's delta was used to identify differentially abundant autoantibodies.
Results: ACPA+ RA patients, ACPA– RA patients, and healthy controls displayed differences in their serum autoantibody profiles (Fig. 1B–D). Upon closer examination, we identified differential serum autoantibodies of ACPA+ and ACPA– RA. Specifically, we found 22 and 19 autoantibodies higher in ACPA+ RA patients and ACPA– RA patients, respectively, compared to healthy controls (Fig. 2A–B). Notably, only one autoantibody for GTF2A2 was found to be higher in both RA subgroups. Next, we found autoantibodies for the CISH protein to be positively correlated with the Clinical Disease Activity Index (ρ = 0.44, P = 6.5 × 10-4) (Fig. 3). Additionally, we determined the functional classes of the proteins targeted by the differentially abundant autoantibodies. We found eight different functional classes, using the PANTHER classification system, for the autoantibodies higher in ACPA+ RA patients, and seven different functional classes for the autoantibodies higher in ACPA– RA patients. When comparing these two groups of functional classes, we identified four in common: 'Gene-specific Transcriptional Regulator', 'Nucleic Acid Metabolism Protein', 'Metabolite Interconversion Enzyme', and 'Protein-modifying Enzyme'. This suggests that an imbalance in autoantibody production targeting metabolic and transcription processes could potentially be a newly identified hallmark of RA.
Conclusion: We identified several differentially abundant autoantibodies in ACPA+ and ACPA– RA, many of which were identified for the first time. Our study demonstrates the potential utility of serum autoantibodies as diagnostic targets for RA subgroups and reveals potential therapeutic treatments.
 Figure 1. Study design overview and group-wise comparisons of autoantibody composition profiles. (A) Blood (serum) samples were collected to investigate serum autoantibody compositions in ACPA+ RA (n = 32), ACPA– RA (n = 30), and healthy controls (n = 30). Each serum sample was screened for 1,622 IgG autoantibodies using the Sengenics KREXTM IMMUNOME Protein Microarray. Raw relative fluorescence units (RFU) values representing serum autoantibody abundances were transformed using quantile normalization prior to all downstream analyses. Heatmap illustrates autoantibodies clustered according to abundance similarities across samples. (B) Principal component analysis on the 92 autoantibody profiles from ACPA+, ACPA–, and healthy controls. Controls showed the highest within-group heterogeneity in autoantibody composition whereas the ACPA+ RA group showed the least within-group heterogeneity. (C) Ternary plot showing mean abundances of 1,622 autoantibodies across ACPA+, ACPA–, and healthy controls. Coordinates of each point correspond to percentages of mean abundances and sum to 100. Despite most autoantibodies having similar mean abundances across the three study groups, several autoantibodies were found to have high abundance in one group (see points closer to the corners). (D) Fold-changes in mean abundances of 1,622 autoantibodies for the RA subgroups compared to the control group. x-axis and y-axis correspond to the fold-changes between ACPA+ RA and controls and between ACPA– RA and controls, respectively. Autoantibodies shown in red points have a fold-change of 2 (or greater) between groups. As observed in (C), several autoantibodies show fold-changes specific to an RA subgroup. The blue diagonal dashed line represents the line y=x.
Figure 1. Study design overview and group-wise comparisons of autoantibody composition profiles. (A) Blood (serum) samples were collected to investigate serum autoantibody compositions in ACPA+ RA (n = 32), ACPA– RA (n = 30), and healthy controls (n = 30). Each serum sample was screened for 1,622 IgG autoantibodies using the Sengenics KREXTM IMMUNOME Protein Microarray. Raw relative fluorescence units (RFU) values representing serum autoantibody abundances were transformed using quantile normalization prior to all downstream analyses. Heatmap illustrates autoantibodies clustered according to abundance similarities across samples. (B) Principal component analysis on the 92 autoantibody profiles from ACPA+, ACPA–, and healthy controls. Controls showed the highest within-group heterogeneity in autoantibody composition whereas the ACPA+ RA group showed the least within-group heterogeneity. (C) Ternary plot showing mean abundances of 1,622 autoantibodies across ACPA+, ACPA–, and healthy controls. Coordinates of each point correspond to percentages of mean abundances and sum to 100. Despite most autoantibodies having similar mean abundances across the three study groups, several autoantibodies were found to have high abundance in one group (see points closer to the corners). (D) Fold-changes in mean abundances of 1,622 autoantibodies for the RA subgroups compared to the control group. x-axis and y-axis correspond to the fold-changes between ACPA+ RA and controls and between ACPA– RA and controls, respectively. Autoantibodies shown in red points have a fold-change of 2 (or greater) between groups. As observed in (C), several autoantibodies show fold-changes specific to an RA subgroup. The blue diagonal dashed line represents the line y=x. Figure 2. Autoantibodies identified to have significantly higher abundances in two subgroups of rheumatoid arthritis (ACPA+ RA and ACPA– RA) than in healthy controls. Patients with (A) ACPA+ RA and (B) ACPA– RA show higher abundances in 22 and 19 autoantibodies, respectively. Two-sided Mann–Whitney U test (P < 0.05) and Cliff’s Delta (|d| > 0.33) were used to find autoantibodies of significantly higher abundance. Standard box-and-whisker plots (e.g., center line, median; box limits, upper and lower quartiles; whiskers, 1.5× interquartile range; points, outliers) are used to show numerical data. *, auto-antibody for GTF2A2 protein was found to have significantly higher abundances in both ACPA+ and ACPA– RA patient groups than in healthy controls.
Figure 2. Autoantibodies identified to have significantly higher abundances in two subgroups of rheumatoid arthritis (ACPA+ RA and ACPA– RA) than in healthy controls. Patients with (A) ACPA+ RA and (B) ACPA– RA show higher abundances in 22 and 19 autoantibodies, respectively. Two-sided Mann–Whitney U test (P < 0.05) and Cliff’s Delta (|d| > 0.33) were used to find autoantibodies of significantly higher abundance. Standard box-and-whisker plots (e.g., center line, median; box limits, upper and lower quartiles; whiskers, 1.5× interquartile range; points, outliers) are used to show numerical data. *, auto-antibody for GTF2A2 protein was found to have significantly higher abundances in both ACPA+ and ACPA– RA patient groups than in healthy controls. Figure 3. Autoantibodies targeting CISH proteins display the highest correlation with Clinical Disease Activity Index (CDAI). The blue line in the scatterplots represents the linear regression fit of Y~X, wherein X and Y correspond to CDAI and autoantibody abundance, respectively. The Spearman correlation coefficient ρ was used to measure the strength of the relationship between the two variables. Scatterplot contains 56 points corresponding to the number of RA patients whose CDAI scores were available.
Figure 3. Autoantibodies targeting CISH proteins display the highest correlation with Clinical Disease Activity Index (CDAI). The blue line in the scatterplots represents the linear regression fit of Y~X, wherein X and Y correspond to CDAI and autoantibody abundance, respectively. The Spearman correlation coefficient ρ was used to measure the strength of the relationship between the two variables. Scatterplot contains 56 points corresponding to the number of RA patients whose CDAI scores were available.Disclosures: K. Cunningham, None; B. Hur, None; J. Davis, Pfizer; J. Sung, None.

