Back

Poster Session D - Tuesday Morning
Category: Biliary/Pancreas
D0014 - Evaluation of a Novel EUS-Compatible Cryoablation Device for the in situ Destruction of Pancreatic Cancer
Tuesday, October 25, 2022
10:00 AM – 12:00 PM ET
Location: Crown Ballroom

Has Audio

John M. Baust, PhD
CPSI Biotech
Owego, NY
Presenting Author(s)
John M. Baust, PhD1, Isaac Raijman, MD2, Anthony Robilotto, PhD1, Robert Van Buskirk, PhD3, Christen Springs, 4, John G. Baust, PhD3, Kristi Snyder, PhD1
1CPSI Biotech, Owego, NY; 2Texas International Endoscopy Center, Houston, TX; 3Binghamton University, Binghamton, NY; 4EndoRX Medical, Houston, TX
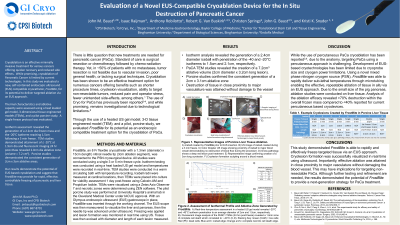
Introduction: There is a pressing need for the development of new devices facilitating advanced minimally invasive approaches for the in situ treatment of pancreatic cancer (PaCa). To this end, a new endoscopic ultrasound (EUS) compatible cryocatheter (Frostbite) has been developed. When paired with the novel Pressurized Subcooled Nitrogen (PSN) cryoconsole, a cryogen is circulated within EUS cryocatheter delivering ultracold ablative temperatures to a targeted tissue in a closed loop manner. In this study, we evaluated cryocatheter performance for its potential use in transesophageal in situ ablation of PaCa and liver cancer.
Methods: A ~1m cryocatheter with a 13cm long 17 gauge needle with a 3cm ablation tip, was connected to PSN and then passed through the working channel of a EUS endoscope. Performance evaluations included a 37°C ultrasound gel model, ex vivo tissue engineered PaCa model and an acute porcine study wherein 6 lesions were created within the liver (under IACUC Approval). Performance assessment included measurement of ice ball size, isotherm profile in real time and destruction area created. A single 5 minute freeze protocol was employed for all evaluations.
Results: Bench studies demonstrated the generation of a 2.4cm diameter iceball with a tip temperature of < -170°C and penetration of the -40 and -20°C isotherms to 1.5cm and 2.1cm (respectively) following 5mins. Analysis of tissue destruction using PaCa tumor model revealed the creation of a 2.1cm ablation area 1 day post freeze. The porcine study demonstrated the consistent generation of a 2cm x 3.1cm (diameter x length) ablation zone following a single 5 minute freeze protocol. The porcine study also demonstrated the ability to deliver targeted destruction of tissue in close proximity to major vasculature without damaging the blood vessel.
Discussion: The results of this study demonstrated that the cryocatheter was able to rapidly and effectively freeze targeted tissue via a EUS approach. The results showed the device was able to consistently ablate a 2cm x 3cm area using a single 5 minute freeze protocol across all models. Analysis of the ablation efficacy revealed ~70% destruction within the overall frozen mass compared to < 40% attained with current percutaneous based cryodevices. Although further testing and refinement are needed, these studies demonstrated the potential of this new approach to provide a next-generation strategy for the treatment of PaCa.
Disclosures:
John M. Baust, PhD1, Isaac Raijman, MD2, Anthony Robilotto, PhD1, Robert Van Buskirk, PhD3, Christen Springs, 4, John G. Baust, PhD3, Kristi Snyder, PhD1. D0014 - Evaluation of a Novel EUS-Compatible Cryoablation Device for the in situ Destruction of Pancreatic Cancer, ACG 2022 Annual Scientific Meeting Abstracts. Charlotte, NC: American College of Gastroenterology.
1CPSI Biotech, Owego, NY; 2Texas International Endoscopy Center, Houston, TX; 3Binghamton University, Binghamton, NY; 4EndoRX Medical, Houston, TX
Introduction: There is a pressing need for the development of new devices facilitating advanced minimally invasive approaches for the in situ treatment of pancreatic cancer (PaCa). To this end, a new endoscopic ultrasound (EUS) compatible cryocatheter (Frostbite) has been developed. When paired with the novel Pressurized Subcooled Nitrogen (PSN) cryoconsole, a cryogen is circulated within EUS cryocatheter delivering ultracold ablative temperatures to a targeted tissue in a closed loop manner. In this study, we evaluated cryocatheter performance for its potential use in transesophageal in situ ablation of PaCa and liver cancer.
Methods: A ~1m cryocatheter with a 13cm long 17 gauge needle with a 3cm ablation tip, was connected to PSN and then passed through the working channel of a EUS endoscope. Performance evaluations included a 37°C ultrasound gel model, ex vivo tissue engineered PaCa model and an acute porcine study wherein 6 lesions were created within the liver (under IACUC Approval). Performance assessment included measurement of ice ball size, isotherm profile in real time and destruction area created. A single 5 minute freeze protocol was employed for all evaluations.
Results: Bench studies demonstrated the generation of a 2.4cm diameter iceball with a tip temperature of < -170°C and penetration of the -40 and -20°C isotherms to 1.5cm and 2.1cm (respectively) following 5mins. Analysis of tissue destruction using PaCa tumor model revealed the creation of a 2.1cm ablation area 1 day post freeze. The porcine study demonstrated the consistent generation of a 2cm x 3.1cm (diameter x length) ablation zone following a single 5 minute freeze protocol. The porcine study also demonstrated the ability to deliver targeted destruction of tissue in close proximity to major vasculature without damaging the blood vessel.
Discussion: The results of this study demonstrated that the cryocatheter was able to rapidly and effectively freeze targeted tissue via a EUS approach. The results showed the device was able to consistently ablate a 2cm x 3cm area using a single 5 minute freeze protocol across all models. Analysis of the ablation efficacy revealed ~70% destruction within the overall frozen mass compared to < 40% attained with current percutaneous based cryodevices. Although further testing and refinement are needed, these studies demonstrated the potential of this new approach to provide a next-generation strategy for the treatment of PaCa.
Disclosures:
John Baust: CPSI Biotech – Employee, Intellectual Property/Patents, Owner/Ownership Interest, Stock-privately held company. GI Cryo – Intellectual Property/Patents, Founder, Owner/Ownership Interest, Stock-privately held company.
Isaac Raijman: Boston Scientific Corporation – Advisor or Review Panel Member, Consultant. ConMed – Speakers Bureau. EndoRx – Owner/Ownership Interest. Endosound – Advisory Committee/Board Member. Medtronic – Speakers Bureau. Microtech – Advisory Committee/Board Member. Pentax – Advisory Committee/Board Member.
Anthony Robilotto: CPSI Biotech – Employee. GI Cryo – Consultant, Intellectual Property/Patents.
Robert Van Buskirk: CPSI Biotech – Employee, Intellectual Property/Patents.
Christen Springs: GI Cryo – Consultant.
John Baust: CPSI Biotech – Advisor or Review Panel Member, Intellectual Property/Patents.
Kristi Snyder: CPSI Biotech – Employee. GI Cryo – Consultant.
John M. Baust, PhD1, Isaac Raijman, MD2, Anthony Robilotto, PhD1, Robert Van Buskirk, PhD3, Christen Springs, 4, John G. Baust, PhD3, Kristi Snyder, PhD1. D0014 - Evaluation of a Novel EUS-Compatible Cryoablation Device for the in situ Destruction of Pancreatic Cancer, ACG 2022 Annual Scientific Meeting Abstracts. Charlotte, NC: American College of Gastroenterology.
