Back

Poster Session C - Monday Afternoon
Category: Biliary/Pancreas
C0011 - Development of a Clinical Screening Tool for Exocrine Pancreatic Insufficiency in Patients With Chronic Pancreatitis
Monday, October 24, 2022
3:00 PM – 5:00 PM ET
Location: Crown Ballroom


Mohamed Othman, MD
Baylor College of Medicine
Houston, TX
Presenting Author(s)
Award: Presidential Poster Award
Mohamed Othman, MD1, Jens Kort, MD, PhD2, Jun Yu, MD, PhD3, Vikesh Singh, MD4, Christopher Forsmark, MD5, Luis Lara, MD6, Walter Park, MD7, Zuoyi Zhang, PhD8, Dhiraj Yadav, MD9
1Baylor College of Medicine, Houston, TX; 2AbbVie, Inc., North Chicago, IL; 3AbbVie Inc., Mettawa, IL; 4Johns Hopkins University, Baltimore, MD; 5University of Florida, Gainesville, FL; 6The Ohio State University Wexner Medical Center, Columbus, OH; 7Stanford University, Stanford, CT; 8AbbVie Inc., North Chicago, IL; 9University of Pittsburgh, Pittsburgh, PA
Introduction: No accurate diagnostic tests exist for exocrine pancreatic insufficiency (EPI). As a result, EPI is misdiagnosed, including in chronic pancreatitis (CP), and there is an unmet need for clinical tools to help clinicians evaluate patients with CP at risk for EPI. The aim of this study was to generate a simple clinical tool to predict EPI in patients with CP.
Methods: 49 variables from medical records of CP patients with or without EPI were entered into a Classification and Regression Tree (CART) model using the unimputed full analysis set and 4 other models (Logistic Regression with Least Absolute Shrinkage and Selection Operator regularization, Support Vector Machine, Random Forest, and Gradient Boosting Machine) using imputed full analysis set. EPI misclassification rate (mRate) served as primary metric to train the 5 prediction models. Each model’s generalizability was assessed using 5-fold cross-validation and the model that was best suited for clinician use based on mRate and other characteristics was selected.
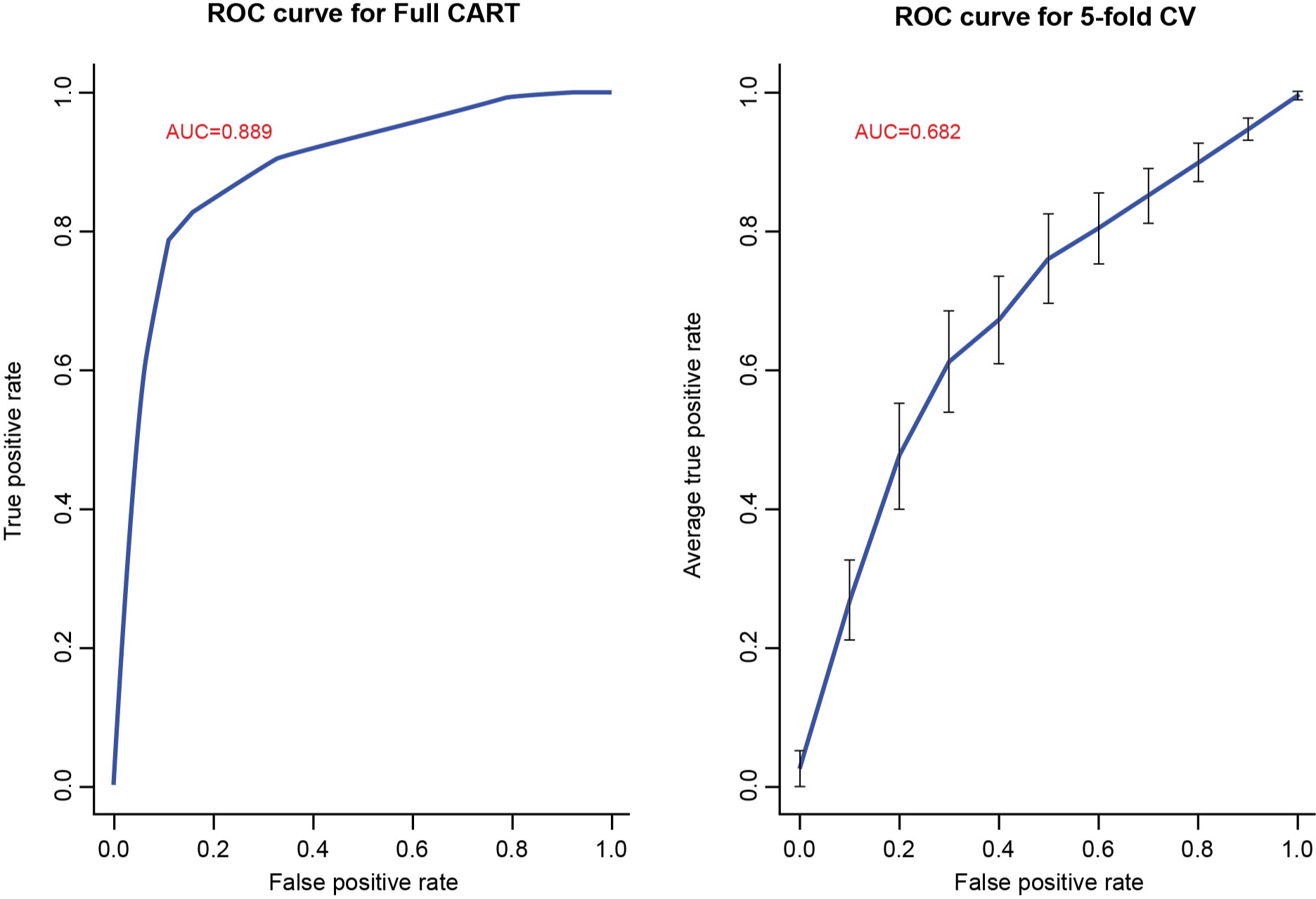
Results: Records of 274 patients with CP from 6 pancreatitis centers across the US were included. Among the 160 (58%)/114 (42%) patients with EPI/without EPI, 48%/55% were female, mean age was 55/52 years, 76%/69% were white, 29%/52% were never smokers, and 39%/53% were never alcohol users. All 5 prediction models demonstrated similar EPI mRates. The CART model was selected. The final CART decision tree includes 10 variables (in order of importance): smoking, pancreatic atrophy, steatorrhea, alcohol use, hemoglobin, serum vitamin D, pancreatic duct contour change, serum albumin, pancreatic parenchymal calcifications, and use of opioid analgesics. The mRate without and with 5-fold cross-validation of the CART was 0.153 (training error) and 0.314 (prediction error), and the area under the receiver operating characteristic curve was 0.889 and 0.682, respectively (Figure 1). Sensitivity/specificity without/with 5-fold cross validation was 0.888/0.789 and 0.794/0.535, respectively.
Discussion: A parsimonious set of 10 variables associated with EPI in CP was identified. The CART decision tree demonstrated good prediction performance for EPI and was selected as the most practical screening tool to aid clinicians in the assessment of EPI in patients with CP. Further external validation of the EPI screening tool is planned.
Disclosures:
Mohamed Othman, MD1, Jens Kort, MD, PhD2, Jun Yu, MD, PhD3, Vikesh Singh, MD4, Christopher Forsmark, MD5, Luis Lara, MD6, Walter Park, MD7, Zuoyi Zhang, PhD8, Dhiraj Yadav, MD9. C0011 - Development of a Clinical Screening Tool for Exocrine Pancreatic Insufficiency in Patients With Chronic Pancreatitis, ACG 2022 Annual Scientific Meeting Abstracts. Charlotte, NC: American College of Gastroenterology.
Mohamed Othman, MD1, Jens Kort, MD, PhD2, Jun Yu, MD, PhD3, Vikesh Singh, MD4, Christopher Forsmark, MD5, Luis Lara, MD6, Walter Park, MD7, Zuoyi Zhang, PhD8, Dhiraj Yadav, MD9
1Baylor College of Medicine, Houston, TX; 2AbbVie, Inc., North Chicago, IL; 3AbbVie Inc., Mettawa, IL; 4Johns Hopkins University, Baltimore, MD; 5University of Florida, Gainesville, FL; 6The Ohio State University Wexner Medical Center, Columbus, OH; 7Stanford University, Stanford, CT; 8AbbVie Inc., North Chicago, IL; 9University of Pittsburgh, Pittsburgh, PA
Introduction: No accurate diagnostic tests exist for exocrine pancreatic insufficiency (EPI). As a result, EPI is misdiagnosed, including in chronic pancreatitis (CP), and there is an unmet need for clinical tools to help clinicians evaluate patients with CP at risk for EPI. The aim of this study was to generate a simple clinical tool to predict EPI in patients with CP.
Methods: 49 variables from medical records of CP patients with or without EPI were entered into a Classification and Regression Tree (CART) model using the unimputed full analysis set and 4 other models (Logistic Regression with Least Absolute Shrinkage and Selection Operator regularization, Support Vector Machine, Random Forest, and Gradient Boosting Machine) using imputed full analysis set. EPI misclassification rate (mRate) served as primary metric to train the 5 prediction models. Each model’s generalizability was assessed using 5-fold cross-validation and the model that was best suited for clinician use based on mRate and other characteristics was selected.
Results: Records of 274 patients with CP from 6 pancreatitis centers across the US were included. Among the 160 (58%)/114 (42%) patients with EPI/without EPI, 48%/55% were female, mean age was 55/52 years, 76%/69% were white, 29%/52% were never smokers, and 39%/53% were never alcohol users. All 5 prediction models demonstrated similar EPI mRates. The CART model was selected. The final CART decision tree includes 10 variables (in order of importance): smoking, pancreatic atrophy, steatorrhea, alcohol use, hemoglobin, serum vitamin D, pancreatic duct contour change, serum albumin, pancreatic parenchymal calcifications, and use of opioid analgesics. The mRate without and with 5-fold cross-validation of the CART was 0.153 (training error) and 0.314 (prediction error), and the area under the receiver operating characteristic curve was 0.889 and 0.682, respectively (Figure 1). Sensitivity/specificity without/with 5-fold cross validation was 0.888/0.789 and 0.794/0.535, respectively.
Discussion: A parsimonious set of 10 variables associated with EPI in CP was identified. The CART decision tree demonstrated good prediction performance for EPI and was selected as the most practical screening tool to aid clinicians in the assessment of EPI in patients with CP. Further external validation of the EPI screening tool is planned.

Figure: Figure 1. AUC-ROC of the CART model trained on the full analysis set and after 5-fold cross-validation. AUC, area under the curve; CART, classification and regression tree; CV, cross-validation; ROC, receiver operating characteristic.
Disclosures:
Mohamed Othman: AbbVie – Consultant, Grant/Research Support. Apollo – Consultant. Boston Scientific Corporation – Consultant. ConMed – Consultant, Grant/Research Support. Lucid Diagnostics – Grant/Research Support. Lumendi – Consultant. Olympus – Consultant.
Jens Kort: AbbVie – Employee, Stock Options.
Jun Yu: AbbVie – Employee, Stock Options.
Vikesh Singh: AbbVie – Consultant, Grant/Research Support.
Christopher Forsmark: AbbVie – Grant/Research Support.
Luis Lara: AbbVie – Consultant, Speakers Bureau.
Walter Park: AbbVie – Advisory Committee/Board Member, Consultant. Acumen – Consultant. Ariel Medicine – Advisory Committee/Board Member, Consultant. Ionis – Advisory Committee/Board Member, Consultant. Nestle – Advisory Committee/Board Member, Consultant. Olympus – Consultant.
Zuoyi Zhang: AbbVie – Employee, Stock Options.
Dhiraj Yadav indicated no relevant financial relationships.
Mohamed Othman, MD1, Jens Kort, MD, PhD2, Jun Yu, MD, PhD3, Vikesh Singh, MD4, Christopher Forsmark, MD5, Luis Lara, MD6, Walter Park, MD7, Zuoyi Zhang, PhD8, Dhiraj Yadav, MD9. C0011 - Development of a Clinical Screening Tool for Exocrine Pancreatic Insufficiency in Patients With Chronic Pancreatitis, ACG 2022 Annual Scientific Meeting Abstracts. Charlotte, NC: American College of Gastroenterology.

